Science Behind the Lesson: It's a Bird! It's a Plane! It's Biomechanics!
Editor: Justin Shaffer
Published online:
Abstract
Active flight is a specialized mode of movement found in insects, birds, and mammals. The anatomy, morphology, physiology, and ecology of flying taxa has changed over evolutionary time. Researchers have proposed several hypotheses about the drivers that fueled the evolutionary transitions necessary to perform flight. Flight is advantageous for animals because it allows them to undergo long and extensive migrations, evade predators, occupy highly specialized niches, and utilize their wing structures in social and mating behaviors. Here we explore the mechanics required to achieve flight. In true flight, a forward moving, airborne body interacts with the following forces: gravity, lift, thrust, and drag. Reynolds number and wing loads dictate speed, ease and efficiency of maneuvering, and spatial constraints of a flying animal in its respective environment. Humans have been inspired by airborne locomotion in nature; aircrafts mimic the form and structure of flying animals. Although insects, birds, and bats have anatomical and physiological differences, they have convergently evolved wing structures that serve as airfoils in order to initiate and maintain active flight.
This article accompanies the lesson "It's a Bird! It's a Plane! It's Biomechanics!"
Citation
Ingle D, Bruellman R, Espana E, Galloway K, Anderson T, Meredith TL, Porter ME. 2019. Science Behind the Lesson: It’s a Bird! It’s a Plane! It’s Biomechanics! CourseSource. https://doi.org/10.24918/cs.2019.43Article Context
Course
Article Type
INTRODUCTION
The following background information is organized into several subsections to describe the physics of flight as well as the hypothesized origins of this specialized mode of locomotion and the biology of organisms that utilize it. First, in forces in flight, principle forces that influence a flying object are described. Next, the major hypotheses regarding the origins and evolution of flight in insects, birds, and mammals are explained. The third major concept details the anatomy of insects, birds, and mammals who share body structures such as wings that aid in active flight. Next, physiology of flying animals explains adaptive energy expenditure and oxygen consumption that support active flight. Finally, connections between flying animals and the specialized niches they occupy are detailed in the ecology of flying animals section.
FORCES IN FLIGHT
There are four principle forces that affect the trajectory of a flying object: gravity, lift, thrust, and drag (Figure 1). The following paragraphs detail the two pairs of opposing forces.
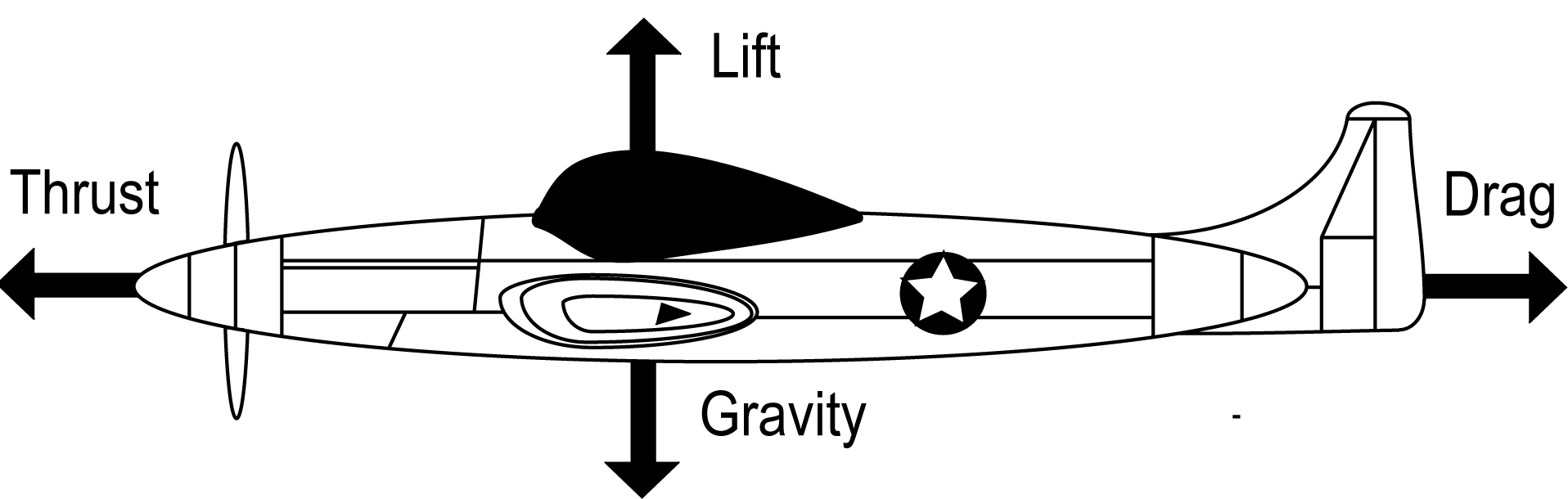
Figure 1. The four principle forces of flight.
Gravity is the attractive force all objects with mass exert on one another (Figure 1). On Earth, gravity is a force that gives weight to objects and pulls flying objects back to the ground. The heavier an object, the more difficult it is to lift it off the ground due to gravity's stronger pull on it. Thus, flying objects must continuously expend energy to counter gravity and remain in the air (1). Lift is the upwards force needed to overcome gravity and leave the ground (Figure 1). Lift can be produced by a variety of mechanisms such as heating air for hot air (balloons) to rise, utilizing a gas less dense than the atmosphere (helium in airships), or via an upwards propulsive force (rockets) where thrust behaves as the lift force. The most common method of producing lift in both animals and aircraft is by utilizing an airfoil to divert oncoming air downwards to create an equal and opposite reaction (lift) upwards. When the lift force on an object is greater than its weight, the object will rise into the air (1,2).
Thrust is the force that propels a flying object forward (Figure 1) and is usually produced by pushing air backwards at a velocity fast enough to push the object forward. This is the force generated by the wings of a bird or by the engines of an aircraft. Thrust is required at speeds where enough air is diverted downwards to produce a sufficient lift force. At insufficient speeds, not enough air is diverted downwards fast enough to produce the appropriate lift force. Without enough lift, the object stalls and gravitational forces pull it to the ground (1,2). Drag opposes thrust and is the hindering force an object generates as a consequence of moving through a fluid (air or water) medium (Figure 1). Drag force is created when an object pushes against the viscosity of a fluid medium. The faster an object moves through the fluid, the stronger the drag force that acts on it. There are two kinds of drag produced as a result: pressure drag and friction drag. Pressure drag (also called parasitic drag) is the result of aerodynamic forces moving at right angles to the surface of an object. The air striking an aircraft's nose directly is an example of pressure drag. Friction drag is produced by air running parallel along the object's body: for example, the air moving around a streamlined aircraft or over and under the wings (1,2).
Airfoils, or structures that provide a reactive force when in motion relative to the surrounding air, are necessary for aircraft and animal flight. Airfoils have a cross-sectional wing shape that provides a large lifting surface with minimal drag. The shape of an airfoil plays an instrumental role in the performance and lift-generating capabilities of an aircraft. The leading edge must be curved to provide minimal drag-inducing resistance from oncoming air while the trailing edge must be sharp to allow air over and under the wing to pass freely. The mechanisms of flight have been traditionally explained with Bernoulli's principle, which states that pressure lowers as air speeds up; a wing generates lift because the air travels faster over the top creating a region of low pressure, and thus lift (3). Bernoulli's principle is further supplemented with the "principle of equal transit times," which states that air separating over the top and bottom of the wing's leading edge converges again at the trailing edge. However, if this were the case, the top surface of a typical small airplane would have to be 50% longer than the bottom edge. The improbability of a universal implementation of this among all aircraft is where this process is challenged by a great body of literature, which has shifted more in favor of Newton's third law, which states that for every action there is an equal and opposite reaction (3). When an aircraft is in forward motion, the airfoil at a positive angle of attack (Figure 2) diverts oncoming air downward over the topside of the wing. This downward acceleration of air causes an equal and opposite upward force on the wing that generates lift. This acceleration of air caused by the airfoil also lowers the air pressure on the topside relative to the higher pressure on the bottom. A pressure gradient over and under the wing is formed also assisting in lift generation (3).
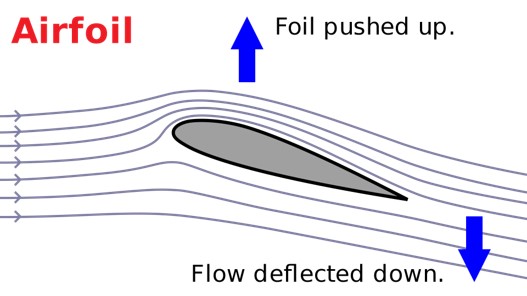
Figure 2. Lift generated on an airfoil by a downward deflection of moving air.
As fluid moves around an airfoil and generates lift, it can also produce trailing vortices. Vortices left in an object's wake are the result of the pressure gradient around the wings that causes high pressure air under the wings to spill over to the low-pressure top. Spill-over occurs at the wing tips and gives the passing air a sideways flow component, disrupting the ideal front-to-back flow of air and producing a downwash behind the object. Trailing vortices create a drag force called induced drag (1,2).
Wing loads are variables calculated by dividing body weight by the total wing area. Objects with high wing loads generally have small wings in relation to a large mass. These smaller wings must work harder and fly faster to produce necessary lift to stay airborne. Objects with a low wing load generally have large wings in relation to a small mass. These larger wings can sustain a light aircraft at lower speeds because they have more surface area for air to move past. High and low wing loads each have strengths and weaknesses for the performance of an object in flight. These include maneuverability, rate of climb, and top speed (Table 1; 1, 2).
The angle of attack is the angle between the relative flow of air and the chord line, or the shortest distance between the front and back of an airfoil (Figure 3). Airfoils have a lift coefficient that varies with the angle of attack.
Lift coefficient (CL) = 2L/ (r * v2 * A) (Equation 1)
Where L =lift; r =density; A =wing area
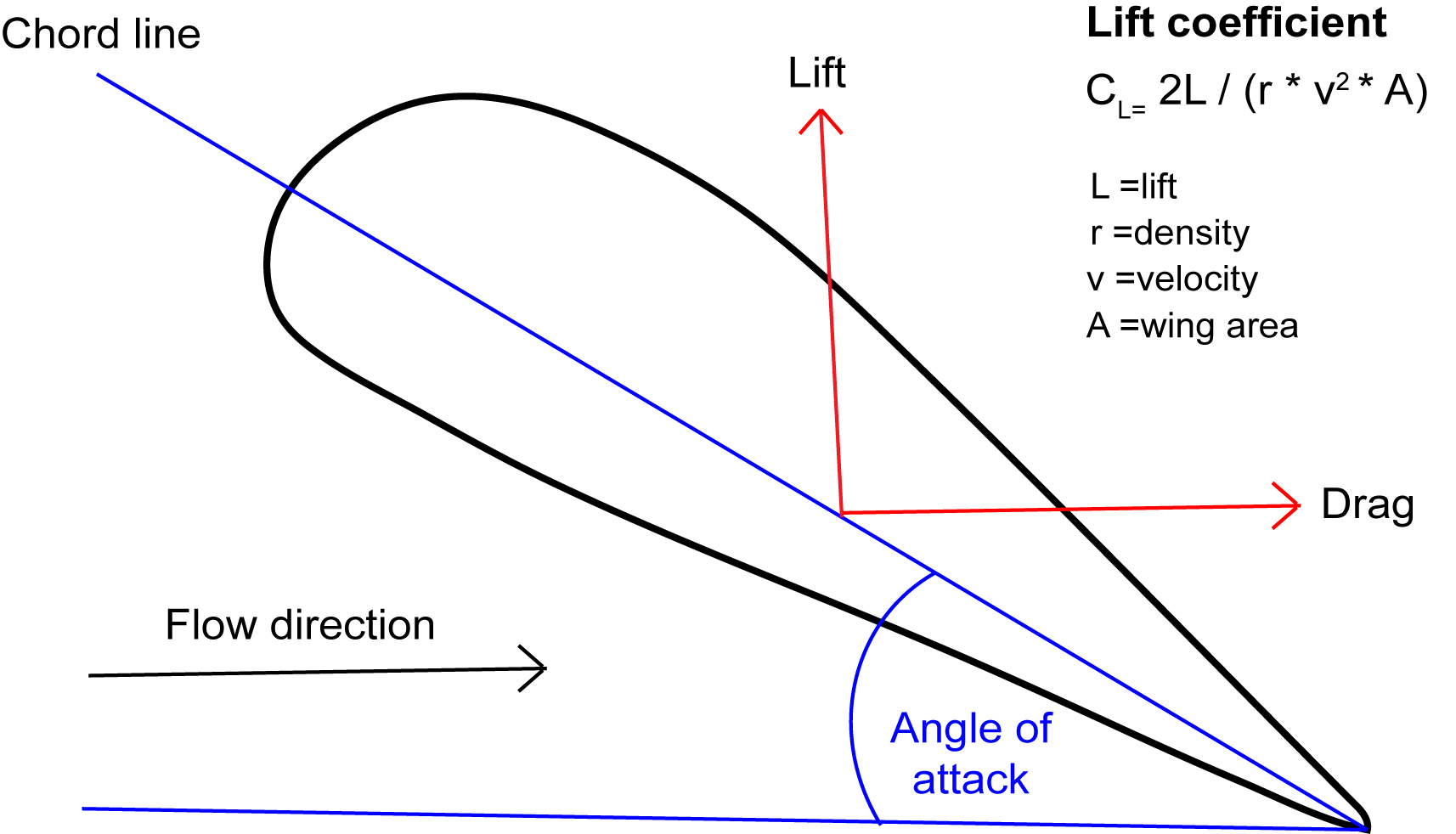
Figure 3. Airfoils have a lift coefficient that will change as the angle of attack changes.
At a zero angle, the ideal flow of air over and under the airfoil occurs. As the angle increases, air traveling over the airfoil must pass an increasing distance to the end. This accelerates oncoming air faster and a stronger lift force is produced. With an increasing angle of attack, the lift coefficient increases to a point. This point is the critical angle of attack, where the maximum lift coefficient for an airfoil is achieved. Once the angle exceeds this value, the ideal laminar flow of air over the wing has been disrupted and lift diminishes until the object stalls. High performance aircraft such as military fighters must have a high critical angle of attack to reliably perform aggressive combat maneuvers (1,2).
EVOLUTION OF FLIGHT IN INSECTS, BIRDS, AND MAMMALS
A series of anatomical, morphological, or physiological shifts in organisms are driven by a combination of genetic mutations and natural selection, and potentially additional various factors, that lead to the evolution of different species. If viable members of a population have a higher fitness due to a certain characteristic, it will likely persist to subsequent generations in a population. Throughout evolutionary history, wings provided certain species with a phenotypic advantage to thrive in environments inaccessible to their earth-bound counterparts. Flight requires a specialized set of physical characteristics, and it is important to understand which evolutionary processes drove flying animals to have such morphologies. Why did groups of insects, birds, and mammals evolve the ability to fly? If we consider the phylogenetic relationship between these animals that are capable of flight, we can see that they have high degrees of separation. Therefore, these taxa underwent convergent evolution, or the process in which organisms not closely related develop similar traits to occupy like environments. The following sections detail hypotheses regarding the appearance of wings, and subsequently powered flight, among taxa.
Insects
In the fossil record, the first winged insects are dated back to the Carboniferous Period (354-298 mya). The following hypotheses list potential origins of wings as precursors to flight in insects: (A) epicoxal hypothesis: wings arose from tracheal gills that were modified for locomotion and expanded into wings (4), (B) paranotal hypothesis: wings developed from paranotal lobes with secondarily-derived innervation and musculature to parachute, glide, and produce active flight (5), and (C) endite-exite hypothesis: inner and outer leg segments extended out to develop laterally projecting wings (6,7).
Birds
To date, Archaeopteryx is the oldest avian fossil (150 mya) to have primary and secondary flight feathers (8). The following hypotheses address potential origin of avian wings and flight: (A) ground-up theory: ancestral species would leap from the ground as they ran, and over generations developed wings and feathers to aid in propulsion, (B) insect net theory: wings were originally used for efficient prey capture that eventually transitioned into powered flapping, and (C) trees-down theory: bird ancestors jumped from tree to tree, and over time development of wings and feathers improved their aerodynamics in gliding and enabled them to eventually perform true flight (8).
Mammals
While many mammals may glide, the only mammalian clade capable of true flight is the bat. Systematic studies based on molecular and paleontological data suggest that the oldest ancestor of the modern bat evolved approximately 64 mya during the Paleocene Epoch (9). One evolutionary model claims that the ancestors of bats initiated powered flight from terrestrial locomotion with a sufficient speed to leap and lift their entire bodies off of the ground (10). However, the thin membrane between the forelimbs and hindlimbs of the bat may produce too much drag, which would hinder its ability to reach cursorial speeds necessary to take off from the ground. Another and more widely accepted model suggests that gliding was most likely the intermediate step between non-flying bat ancestors and bats capable of powered flight (11,12).
ANATOMY OF INSECTS, BIRDS, AND MAMMALS
Flying insects, birds, and mammals have convergently evolved their body plans in order to engage in true flight. While they share the characteristic of having wings, these taxa have high degrees of phylogenetic separation and therefore show distinct morphologies to facilitate flight.
Insects
Insect wings lack muscle control over the entire length of the wing and are supported by a stiff vein network. Muscles that control the wing are located proximally at the base where the wing is attached to the insect body (13), and these flight muscles are a major component of the body's musculature. Insects that beat their wings less than 100 times per second use a synchronous muscle (beats once per nerve impulse) and insects that beat their wings for more than 100 times per second use an asynchronous muscle (beats more than once per nerve impulse; 14). Given the lack of control insects have over manipulating their wing structure, insect wings are known to be much more rigid when compared to bats or most bird wings. Similar to bats and birds, different wing ratios are present among insect species. Some insects such as dragonflies possess long wings, while others such as butterflies have shorter and broader wings. Dragonflies, with their higher aspect ratio wings, can sustain longer hovering flight compared to butterflies. Some insect wings are paired, which can further assist them with providing more thrust in flight, while other species have one pair of wings similar to a bird or a bat.
Birds
The bird wing has three fused digits that form moderately inflexible wings, although there is often a higher degree of flexibility compared with insect wings. The bird's large pectoralis and supracoracoideus muscles power the wings in the downward and upward flight strokes, respectively (Figure 4; 15). A distinguishing characteristic of bird wings is the feather, which serves as a lightweight insulator and aerodynamic aid in flight. Birds also sport hollow bones and a lightweight beak to facilitate flight.
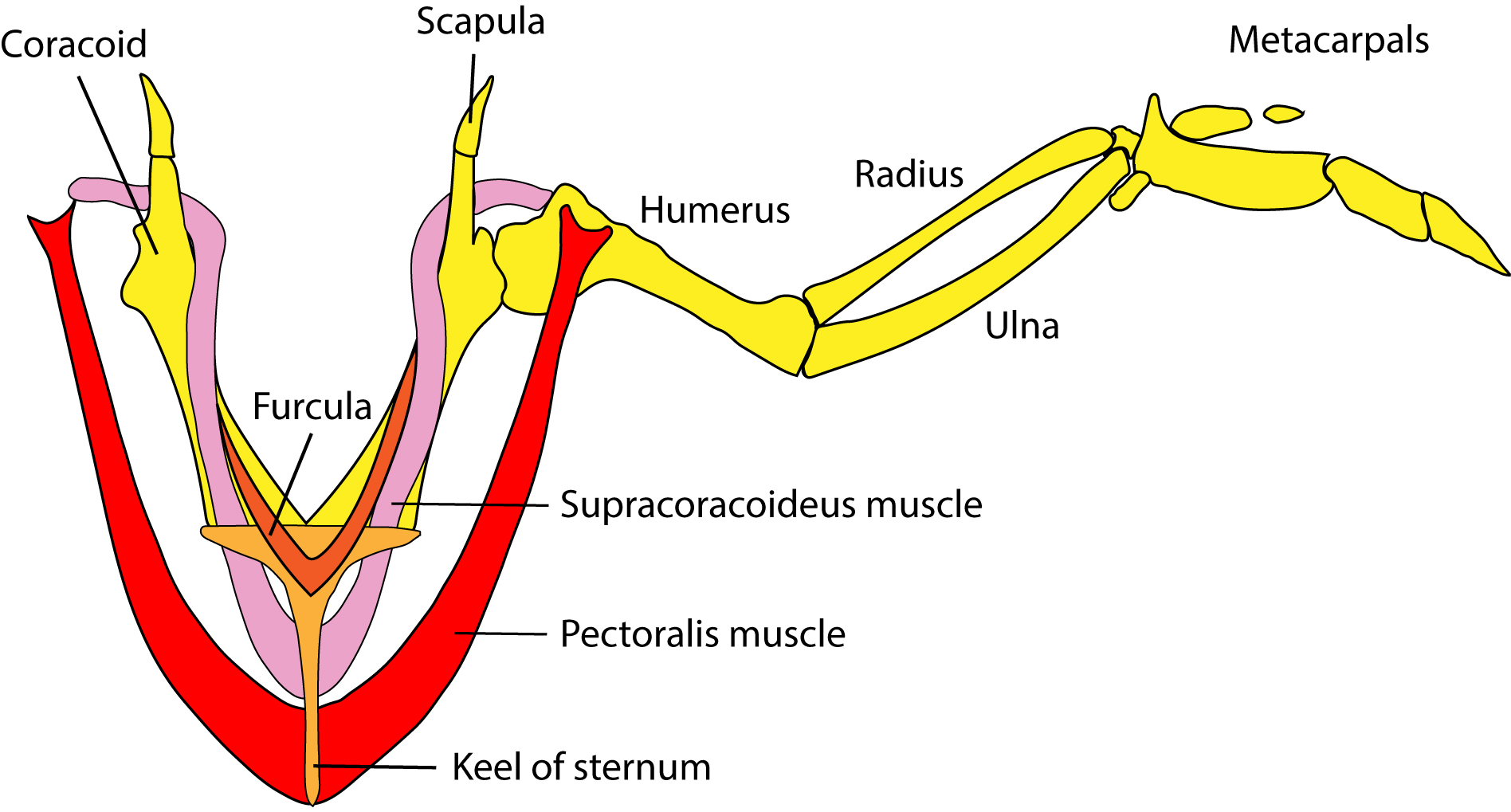
Figure 4. Musculoskeletal anatomy of a bird wing.
In tandem with shape and physical characteristics of the wing, a key component for bird flight is how the wing is angled. For forward flight, it is optimal for the wing to be angled slightly upwards in order to create a pressure gradient to counter the force of gravity (16). A bird's aspect ratio (the ratio of width to height) is an important indicator of how it flies, and this ratio dictates how birds can effectively navigate their respective environments. In many birds, there are distinct intraspecies differences found in flying anatomy. One study found anatomical differences in a species of sparrow depending on whether the population resided in a low- or high-altitude habitat (17). Higher altitude sparrows, for example, had longer and more rounded wingspans compared to those at lower altitudes. This morphological deviation is attributed to the need to meet physical demands of maneuvering through thinner air found at high altitudes. In addition, lung and heart size were found to be larger in the higher altitude sparrows to adjust to lower air pressure and atmospheric oxygen (17).
Mammals
The only mammal able to engage in powered flight, the bat, has wings homologous to birds based on a similar location of the body, skeletal structure, the number of wings, and the same general function. However, bats have a unique wing system compared to birds or insects. A bat wing includes bones that form the limbs and digits with a very thin membrane stretched between the bones. Each of the five phalanges, as well as the humerus and radius arm bones, are slim and extended compared to most other mammalian forelimbs. This allows for a larger membrane area, more flexibility, and a larger area of wings. Each of the digits has numerous joints, which contribute to overall flexibility of the wing (18), and the wing itself can change shape with each beat. During flight, contracting muscles tighten excess membrane to reduce additional drag on the bat (19). The posterior end of the bat has the uropatagium, which is a thin membrane stretched between the legs. This structure varies in size among bat species and is thought to assist with steering as a bat flies (18). Different bat species have various and distinctive wing aspect ratios and wing tip lengths. Extended, long wings with shorter tips are generally associated with bats that are slower in speed but can maintain their position in the air longer. Bats having short and wide wings (i.e., low aspect ratio) excel in ecological niches where they can maneuver through heavy vegetation. Similarly, bats with a high aspect ratio (long and narrow wings) benefit in open areas that are conducive to a wide wingspan (20). Bats with low wing loads (lower body weight relative to wing area) have been shown to maneuver well through crowded forests and can carry relatively heavy prey while flying; by contrast, bats with high wing loads (higher body weight relative to wing area) are able to reach higher velocities (21).
PHYSIOLOGY OF FLYING ANIMALS
The fuel for a long-distance flight comes primarily from stored fat, a major contributor to overall body mass. Maximum flight distance has been shown to correlate with body mass, where maximum flight distance equals body mass 0.227 (22).
Flight requires an extremely high-power output (work / time); however, flying is an energetically cheaper and faster way to move than running. Furthermore, gliding and soaring compared to powered flight are even less energetically costly ways for animals to move about as they forage for resources. For example, the peregrine falcon is the fastest organism in the animal kingdom; it soars to steep heights in order to dive at speeds of over 200 mph (23). In insects, some of which have asynchronous flight, it has been shown that the metabolic rate does not vary with flight speed compared to their hovering speed; therefore, this taxon can reach top speeds without much additional energetic cost (24). This is contrary to the long held assumption that all flying animals and insects fit the same mold of having an optimal intermediate flight speed with slower and faster speeds resulting in higher energetic cost. While this assumption does hold for many animals such as birds and bats, flying insects do not hold to the assumption.
Unlike mammals, bird respiration requires the unidirectional flow of oxygen through specialized air sacs. This flow permits higher oxygen content to enter the lungs with each breath as it does not mix with air that has been already in the lungs and has a lower oxygen content. Adaptive air sacs allow birds to take in oxygen even during exhalation so they can breathe at higher elevations when flying (25). Birds also lack diaphragms and move air in and out of their respiratory system through pressure changes in the air sacs. The enlarged pectoralis muscles cause the sternum to be pushed outward and this creates a negative pressure in the air sacs, causing air to enter the respiratory system. Muscles then contract to increase the pressure on the air sacs and push the air up and out through the trachea (25). Based on the higher VO2 (oxygen consumption) and lower respiratory rate found in birds when compared to mammals of similar sizes, birds spend less energy breathing for the same gains in oxygen. Thus, bird respiration is thought to have evolved in climates where oxygen was very limited (23,25). The metabolic cost of powered flight in birds and bats are similar, and the VO2 of bats has been shown to increase by 30 times in flight when compared to a state of rest. Flying bats have a high hematocrit and oxygen carrying capacity in their blood as an adaptation to maintain such high metabolic rates during flight (26).
ECOLOGY OF FLYING ANIMALS
Powered flight offers a series of advantages that are otherwise unavailable to earth-bound animals: extensive migrations, predator evasion, and highly specialized niche occupation. Although wings and powered flight are not mutually inclusive in all species (some have wings but cannot fly), animals that perform true flight also use their wings to engage in social and mating behaviors unrelated to flight. In birds, insects, and bats, winged appendages have allowed tremendous specializations that enhance animal fitness.
Annual migrations occur in numerous taxa, and flying insects, birds, and bats can cover great distances to access resources or reproduce. Migrating insects are often heavily influenced by wind and save energy by flying via downwind gusts to disperse across a terrain (27). Like insects, bird migration is subject to wind factors such as speed and temperature, which determine the altitude and routes of migration (28,29). Certain birds will nest and fledge (young learn to fly) in one region and then fly hundreds of miles for a new climate and set of resources (30). This strategy prevents overuse of nutrients at nest sites and gives fledglings the ability to strengthen their wings and flying skills over long journeys. Biomechanically, many species of birds utilize intermittent flight for long range journeys, a variation between lift-generating flapping and lift maintaining gliding. This behavior saves energy and keeps them at maximum range velocity. Conversely, short range fliers can use intermittent flight to reduce energy output by alternating between flapping and bounding, which are pauses between flapping where the bird maintains a flexed wing to maintain altitude. In bats, researchers have found that migrations are generally sex-biased; females migrate more than males, which is not commonly found in birds (31).
Flying animals tend to build nests in high, secluded areas (32,33). Examples include bees and wasps that create high-placed cavernous nests and dragonflies that adhere their eggs to foliage protruding from aquatic areas. All of these strategies add an element of difficulty or inaccessibility for predators (33). Bird nests and bat roosts can be variable in placement and composition; however, almost all of them are either in high locations or are heavily vegetated. Once outside of the nest, there are additional predators for flying animals to avoid. One advantage flying insects have is that they are difficult to capture; they dart around at speeds which the human eye has difficulty following (34). Conversely, terrestrial insects are at a tremendous disadvantage when fleeing predators due to their small size, which limits the ground they are able to cover during an escape (34).
The opportunities for resource partitioning, or the division of resources among several species for mutual advantage, expand greatly for animals capable of flight. These insects, birds, and bats are able to exploit resources that are often out of reach or inaccessible for their earth-bound counterparts. Most insects feed on live foliage, fruit, or decaying matter, and flying insects are able to lower feeding competition by selecting highly-placed vegetation (34). Hummingbirds feed on the nectar of delicate flowers that are unable to support the bird's weight by hovering for long periods of time (35). Woodpeckers also minimize their competition for food by using their beak as a jackhammer to access insects within tree trunks that are shielded from most other predators (36). Bat species partition resources based on their diet and include frugivores, carnivores, nectarivores, piscivores, and sanguivores (37). Other animals, such as seabirds, divide their time, energy, and resources between terrestrial and aquatic habitats. These animals often nest on land and hunt underwater and are well known for their air-to-sea transitions; progressing from powered flight to a highly streamlined nose dive into the water to capture prey (38). In the face of urbanization, many flying species have partitioned where they make their homes; some remain in purely natural areas such as forests and caves, while others have adapted well to living in urban, man-made structures (39,40).
Some birds and insects use wing displays to attract mates or intimidate conspecifics (individuals of the same species) to defend their territory. Drosophila males have been shown to exhibit mating behaviors such as vibrating, flicking, waving, and scissoring of their wings (41). Other species also rely on wing color to attract and locate mates, such as the Helioconius butterflies (42). One study observed and described how male finches raise and fluff their wings as a sexual display to attract females (43).
CONCLUSION
Wings arose over millions of years of evolutionary history and were coupled with morphological and physiological shifts as animals radiated into their respective environments. Gravity, lift, thrust, and drag are the principle forces that facilitate flight, while wing load and angle of attack contribute to speed and maneuvering. Wings are analogous structures shared by insects, birds, and bats; they arose from different evolutionary origins and consist of different tissues among taxa, but provide the same overall function. Various physiological adaptations help many of these animals to thrive in high altitudes or complete long, airborne migrations. Flight facilitates niche partitioning, allowing animals to access highly-placed and cavernous food sources otherwise unreachable to their earth-bound counterparts. In addition to the mechanical function of wings, they can also serve as magnificent displays for mating and sexual selection. Numerous man-made structures are mechanically reminiscent of animal wings, and we continue to draw from their diverse morphologies to develop new technologies.
ACKNOWLEDGMENTS
We would like to thank A.D. Henderson University School for granting us the opportunity of sharing our lesson plan with their students.
References
- Alexander R. 2013. Powered forward flight, p. 224-239. In Principles of Animal Locomotion. Princeton University Press, Princeton.
- Yechout T. 2003. Introduction to aircraft flight mechanics. Reston, VA. AIAA. doi:10.2514/4.862069
- Anderson D, Eberhardt S. 2015. How airplanes fly: A physical description of lift. Retrieved from http://www.aviation-history.com/theory/lift.htm
- Woodworth CW. 1906. The wing veins of insects, p. 1-152. In Entomology. University of California Publishing: Technical Bulletin, Berkeley.
- Wootton R. 1976. Insect Flight Symposia of the Royal Entomological Society of London. Blackwell Scientific Publications, London.
- Kukalova-Peck J. 1983. Origin of the insect wing and wing articulation from the arthropodan leg. Can J Zool 61:1618-1669. https://doi.org/10.1139/z83-217
- Trueman J. 1990. Evolution of insect wings: A limb exite plus endite model. Can J Zool 68:1333-1335. https://doi.org/10.1139/z90-200
- Gatesy SM. 2002. Locomotor evolution on the line to modern birds. In Chiape LM, Whitmer LM (eds.), Mesozoic birds: above the heads of dinosaurs (pp. 432-447). University of California Press.
- Teeling E, Springer M, Madsen O, Bates P, O'Brien S, Murphy W. 2005. A molecular phylogeny for bats illuminates biogeography and the fossil record. Science 307 (5709):580-584. https://doi.org/10.1126/science.1105113
- Caple G, Balda R, Willis W. 1983. The physics of leaping animals and the evolution of preflight. Am Nat 121:455-476. https://doi.org/10.1086/284076
- Norberg U. 1990. Vertebrate flight: Mechanics, physiology, morphology, ecology, and evolution. Springer-Verlag, Berlin Heidelberg.
- Simmons J, Saillant P, Wotton J, Haresign T, Ferragamo M, Moss C. 1995. Composition of biosonar images for target recognition by echolocating bats. Neural Networks 8:1239-1261. https://doi.org/10.1016/0893-6080(95)00059-3
- Combes SA. 2003. Flexural stiffness in insect wings II. Spatial distribution and dynamic wing bending. J Exp Biol 206:2989-2997. doi: 10.1242/jeb.00524
- Dudley R. 2000. The biomechanics of insect flight: Form, function, fvolution. Princeton University Press, Princeton. http://dx.doi.org/10.1515/9780691186344
- Tobalske BW. 2007. Biomechanics of bird flight. J Exp Biol 210:3135-3146. doi: 10.1242/jeb.000273
- Iriarte-Diaz J, Riskin D, Breuer K, Swartz S. 2012. Kinematic plasticity during flight in fruit bats: Individual variability in response to loading. PLoS One. https://doi.org/10.1371/journal.pone.0036665
- Sun Y, Ren Z, Wu Y, Lei F, Dudley R, Li D. 2016. Flying high: Limits to flight performance by sparrows on the Qinghai-Tibet Plateau. J Exp Biol 219:3642-3648. doi: 10.1242/jeb.142216
- Hedenstrom A, Johansson LC. 2015. Bat flight: Aerodynamics, kinematics and flight morphology. J Exp Biol 218:653-663. doi: 10.1242/jeb.031203
- Hedenström A, Johansson LC, Wolf M, von Busse R, Winter Y, Spedding GR. 2007. Bat flight generates complex aerodynamic tracks. Science 316:894-897. doi: 10.1126/science.1142281
- Findley JS. 1972. Phenetic relationships among bats of the genus Myotis. Syst Biol 21:31-52. https://doi.org/10.1093/sysbio/21.1.31
- Norberg UM, Rayner JM. (1987). Ecological morphology and flight in bats (Mammalia; Chiroptera): Wing adaptations, flight performance, foraging strategy and echolocation. Philosophical Transactions of the Royal Society of London. B, Biological Sciences, 316(1179), 335-427.
- Tucker VA. 1971. Flight energetics in birds. Am Zool 11:115-124. https://doi.org/10.1093/icb/11.1.115
- Norberg U. 2012. Vertebrate flight: Mechanics, physiology, morphology, ecology and evolution. Springer Science and Business Media, Berlin Heidelberg.
- Ellington C, Machin K, Casey T. 1990. Oxygen consumption of bumblebees in forward flight. Nature 347:472-473. https://doi.org/10.1038/347472a0
- West J, Watson R, Fu Z. 2007. The human lung: Did evolution get it wrong? Eur Respir J 29:11-17. doi: 10.1183/09031936.00133306
- Thomas S, Suthers R. 1972. The physiology and energetics of bat flight. J Exp Biol 57:317-335.
- Gatehouse A. 1997. Behavior and ecological genetics of wind-borne migration by insects. Annu Rev Entomol 42:475-502. https://doi.org/10.1146/annurev.ento.42.1.475
- Pennycuick C. 1975. Mechanics of flight. Avian Biol 5:1-75. http://dx.doi.org/10.1016/B978-0-12-249405-5.50009-4
- Richardson W. 1976. Autumn migration over Puerto Rico and the western Atlantic: A radar study. Ibis (Lond 1859) 118:309-332. https://doi.org/10.1111/j.1474-919X.1976.tb02023.x
- Odum E, Connell C, Stoddard H. 1961. Flight energy and estimated flight ranges of some migratory birds. Auk 78:515-527. doi: 10.2307/4082185
- Tobalski BW. 2007. Biomechanics of bird flight. J Exp Biol 210: 3135-3146. doi: 10.1242/jeb.000273
- Fleming T, Eby P. 2003. Ecology of bat migration, p. 156-208. In Kunz, T, Fenton, M (eds.), Bat Ecology. The University of Chicago Press, Chicago and London. http:// www.press.uchicago.edu/cgi bin/hfs.cgi/00/15369.ctl. ISBN 0-226-46206-4
- Finch D. 1983. Seasonal variation in nest placement of Abert's Towhees. Condor 85:111-113. doi: 10.2307/1367905
- Heinrich B. 1976. Resource partitioning among some eusocial insects: Bumblebees. Ecology 57:874-889. https://doi.org/10.2307/1941054
- Brown B. 1992. Nesting chronology, density, and habitat use of black-chinned hummingbirds along the Colorado River, Arizona. J F Ornithol 393-400. https://sora.unm.edu/sites/default/files/journals/jfo/v063n04/p0393-p0400.pdf
- Beal F. 1911. Food of the woodpeckers of the United States. Biological Survey. http://dx.doi.org/10.5962/bhl.title.55219
- Aguirre L, Herrel A, van Damme R, Matthysen E. 2002. Ecomorphology analysis of trophic niche partitioning in a tropical savannah bat community. Proc R Soc London B Biol Sci 269:1271-1278. https://doi.org/10.1098/rspb.2002.2011
- Sekercioglu C. 2006. Increasing awareness of avian ecological function. Trends Ecol Evol 21:464-471. https://doi.org/10.1016/j.tree.2006.05.007
- Marzluff J, DeLao J, Oleyar M, Whittaker K, Gardner B. 2016. Breeding dispersal by birds in a dynamic urban ecosystem. PLoS One 1-20. https://doi.org/10.1371/journal.pone.0167829
- Jung K, Kalko E. 2011. Adaptability and vulnerability of high flying Neotropical aerial insectivorous bats to urbanization. Divers Distrib 17:262-274. https://doi.org/10.1111/j.1472-4642.2010.00738.x
- Spieth H. 1974. Courtship behavior in Drosophila. Annu Rev Entomol 19:385-405. http://dx.doi.org/10.1146/annurev.en.19.010174.002125
- Kronforst M, Young L, Kapan D, McNeely C, O'Neill R, Gilbert L. 2006. Linkage of butterfly mate preference and wing color preference cue at the genomic location of wingless. PNAS 103:6575-6580. https://doi.org/10.1073/pnas.0509685103
- Hinde R. 1956. A comparative study of the courtship of certain finches (Fringillidae). Ibis (Lond 1859) 98:1-23. https://doi.org/10.1111/j.1474-919X.1956.tb03027.x
Article Files
Login to access supporting documents

Comments
Comments
There are no comments on this resource.