Exploring Miracle Fruit: An Undergraduate Laboratory Exercise on Experimental Design
Editor: Neena Grover
Published online:
Abstract
In recent years, undergraduate biology and biochemistry curricula have seen an increase in the use of course-based undergraduate research experiences (CUREs). CUREs maximize potential student gains when students take an active role in experimental design. Here we propose an activity that can either complement CUREs or function as a stand-alone activity that develops students’ abilities to design an experiment. While most active-learning interventions are designed for the purpose of teaching content, with experimental design skills as a secondary concern, this activity was created primarily to develop experimental design skills, while concurrently teaching important biochemistry concepts. The activity, designed to occur during a single three-hour lab meeting, allows students to experimentally explore the mechanisms of the taste-altering miraculin protein, found in the fruit of Richadella dulcifica, commonly known as miracle fruit. Students in an advanced biology class reported increased understanding of important experimental design concepts and increased knowledge of receptor binding and structural dynamism of proteins. Students also reported learning the importance of identifying nested variables that are difficult to tease apart, particularly when resources, time, or subjects are limited. While intended to develop experimental design skills in an upper-level undergraduate biology course, instructors can adapt the activity to suit biochemistry and introductory biology courses.
Primary image: Miracle Fruit: The image depicts the fruit of Richadella dulcifica, commonly known as Miracle Fruit. This fruit contains the pH-dependent, taste-modifying miraculin protein, which is the subject of this laboratory exercise. https://en.wikipedia.org/wiki/Synsepalum_dulcificum#/media/File:MiracleBerry.jpg (Public Domain)
Citation
Ganser SJ, Hines JK, Butler MW. 2021. Exploring Miracle Fruit: An Undergraduate Laboratory Exercise on Experimental Design. CourseSource. https://doi.org/10.24918/cs.2021.29Society Learning Goals
Biochemistry and Molecular Biology
- Macromolecular Structure Determines Function and Regulation
- How are structure and function related?
- How is macromolecular structure dynamic?
Lesson Learning Goals
- Students will develop skills to confront the obstacles that are an inherent part of designing an objective experiment
- Students will be exposed to the dynamic nature of macromolecules
Lesson Learning Objectives
Students will be able to:- propose a testable hypothesis when given a problem.
- design appropriate data collection methods to test a hypothesis.
- work cooperatively to complete a task and report results.
- identify potential confounds when designing an experiment.
- articulate the role that biochemistry plays in human physiology.
Article Context
Course
Article Type
Course Level
Bloom's Cognitive Level
Vision and Change Core Competencies
Vision and Change Core Concepts
Class Type
Class Size
Audience
Lesson Length
Pedagogical Approaches
Principles of How People Learn
Assessment Type
Introduction
Undergraduate biology educators take on the challenge of building a strong scientific foundation for their students. For some students, however, an introductory biology class will be their only exposure to collegiate science and therefore a commonly-shared goal among most instructors is to develop students’ abilities to think scientifically. This ability, which is critical in promoting a scientifically-literate society, is something that requires expertise in experimental design (1). In recent years, the community of undergraduate instructors has taken steps to prioritize experimental design education (2). Many instructors have developed activities that integrate experimental design concepts, but biological content remains the primary learning goal (1, 3, 4, 5). In order to truly instill a deep understanding of how to conduct rigorous scientific studies, instructors would benefit from biology-content related activities that prioritize experimental design skills as their primary learning goals (4).
Traditional lab exercises can either create, or fail to correct, undergraduate misconceptions about the scientific process (6). For example, undergraduates severely underestimate the importance of social discourse in the generation of scientific knowledge and often mischaracterize the relationship between data and scientific knowledge claims following participation in basic procedural labs (6). Course-based undergraduate research experiences (CUREs), on the other hand, address issues associated with traditional labs by allowing undergraduates to develop crucial experimentation skills while engaging them in the scientific process through authentic research projects (7). However, despite their growing popularity, a significant challenge associated with the CURE approach is that students enter courses with varying levels of prior experience with research and experimental design and thus not all students have the appropriate background to derive maximum benefit from CUREs (8). Most CUREs are characterized by “legitimate peripheral participation,” in which instructors continue to ask the overarching research questions; students still learn valuable skills, including formulating hypotheses, data collection, data analysis, and communication of results, but they are not asking their own research questions (9).
Here we present an activity that we designed and implemented to teach experimental design and that can also be used to help students navigate the gap between the level of inquiry required for traditional undergraduate laboratory exercises and CUREs. The activity strives to dispel common misconceptions about experimental design by providing students with the opportunity to ask authentic research questions and to design and refine experiments to answer them. In doing so, students encounter complexities and challenges that must be overcome to improve the objectivity of their experiments. They also receive real-time, verbal instructor feedback while they plan their experiments, which enables them to reflect on appropriate design processes.
Our activity utilizes a model that both piques student interest and curiosity and grants the opportunity to genuinely engage with an area of science in which many unanswered questions remain. In his 2013 paper, “Molecular mechanisms of the action of miraculin, a taste-modifying protein,” Takumi Misaka details what is known of the mechanisms of the protein miraculin, found in the fruit of Richadella dulcifica, commonly known as “miracle fruit” (10). When eaten, miraculin binds to the human sweet taste receptor and temporarily alters gustation, particularly the perception of acidic (sour-tasting) foods (10). The time period of this fully reversible phenomenon exhibits interpersonal variability, but usually lasts for about 30 minutes following fruit consumption (10). Misaka characterized the relationship between taste perception and pH after miraculin binding (10). Foods of low pH, many of which normally taste sour, result in a strong sweet taste when eaten after miracle fruit (10). Foods of neutral or basic pH, however, taste less sweet than they do in the absence of miraculin, or exhibit no change in taste (10). Misaka proposes that miraculin acts as an antagonist in neutral and basic pH environments, thus blocking the sweet taste receptors but not triggering any cellular process (10). However, in an acidic environment, the shape of the miraculin protein changes, and it begins to behave as an agonist, resulting in a cellular process that triggers sweet taste perception (10). The molecular mechanisms of this process are not yet known, nor are students typically aware of miraculin or its pH sensitivity, making this model ideal for teaching experimental design.
Miraculin has been used to teach undergraduates about taste and sensory perception, and has proved to be an engaging teaching tool (11). However, in that teaching intervention, the primary goal was to teach neuroscience content rather than experimental design and so students followed a predetermined procedure, rather than freely designing their own experiments (11). Here, we set a different goal. Students use the characteristics of miraculin described by Misaka to conduct their own investigation and answer a question of their choosing, thus creating an authentic experimental experience (10).
In our activity, students are placed into groups of three to four individuals and given miraculin and foods of various taste profiles and pH values. They are asked to design an experiment within their groups that will allow them to assess some aspect of how eating the fruit affects their perception of taste. In order to highlight the importance of discourse to the scientific process, students are encouraged to collaborate with other groups and discuss their ideas throughout the design and refinement process. Additionally, the instructor provides feedback during, rather than after, the activity. The mechanisms of miraculin provide students with a variety of variables that they can choose among for testing. For example, some students choose to assess the taste perception of a single food at various time intervals after eating miracle fruit. Other students investigate the effects of miraculin on the tastes of foods of different pH values. The possibilities range far beyond these two examples. No matter what experiment students design, the significant interpersonal variability and difficulty in controlling various aspects of miraculin functions enables them to experience obstacles in their experimental design. For example, students often discover, with help from the instructor, that they are incorrectly quantifying taste, or that they are failing to control for one or more variables, such as interpersonal differences or order effects. The miraculin model allows students to complete the steps for designing and conducting a legitimate experiment, while promoting excitement and engagement through its sensory and unique subject matter.
Intended Audience
This activity was used in an upper-level biology course at a selective, undergraduate-only liberal arts college in the mid-Atlantic United States. Immediately following completion of the activity, 46 students were surveyed. Of the students in the sample who reported their class year, 93% were in their fourth year of college, 4.4% were in their third year, and 2.2% were in their second year. Of those who reported their major, 69% were specifically biology majors, and 98% had majors in STEM fields. The intended audience however is undergraduates at any level of progression and both STEM and non-STEM majors. The activity is appropriate for Introductory Biology, Zoology, Physiology, Biochemistry, or other similar laboratory courses.
Required Learning Time
The lesson was completed during a single, three-hour lab session. Misaka’s “Molecular mechanisms of the action of miraculin, a taste-modifying protein” was provided to students one week prior to the lab activity (10). The students were asked to read the article prior to the lab. The activity was completed during the second week of the semester. It can be adapted to fit longer or shorter class periods, as discussed below.
Prerequisite Student Knowledge
Interpreting a Scientific Article
Before they participate in this lesson, students must be able to successfully obtain key information from a basic scientific article. They will need to carefully read “Molecular mechanisms of the action of miraculin, a taste modifying protein” by Takumi Misaka in order to obtain the necessary background information on miraculin (10); note that this article may require purchase or acquisition via interlibrary loan. In order for students to properly evaluate the results presented in the scientific article, they must approach the article with sufficient basic knowledge and vocabulary to make meaning from it (12). Therefore, students should approach Misaka 2013 with understanding of the vocabulary terms “agonist” and “antagonist” (10). Additionally, students will be most successful if they have some familiarity with the relationship between protein structure and function. They will gain maximum insight from the article if they can define and depict a “signaling cascade.” After reading, students should understand that the miraculin protein binds to the human sweet taste receptor and that its effect on taste is dependent on the pH of its environment, i.e., the food eaten (10).
Experimental Design Skills
Before students participate in this lesson, they should be able to articulate ideas about the goals and process of experimental design. These ideas need not be refined nor detailed. Most importantly, students should be able to recognize a scientific question or hypothesis as testable prior to beginning the activity. Beyond this, students benefit from an understanding of typical features of an experimental procedure (multiple trials, independent and dependent variables, controlled variables, etc.) and ability to communicate ideas and results either verbally or in writing. However, experimental design skills will be refined during the activity, so students need not enter with any further depth of understanding on this topic.
Prerequisite Teacher Knowledge
We recommend that the instructor reads and is familiar with information on protein structure and function, pH, pH-dependent conformational change, receptor binding, and signaling cascades at the level generally presented in undergraduate biology textbooks. Most importantly, since the focus of this activity is the experimental design process, the instructor would benefit from a very strong grasp of experimental design concepts such development of a testable scientific question, experimental controls, data collection methods, and limitations of data. Additionally, they will find it useful to have an awareness of the problems that students frequently face when designing their experiments for the activity. This information can be found in Supporting File S2: Miracle Fruit: Probing Questions to Uncover Experimental Design Issues.
A thorough reading of Takumi Misaka’s “Molecular mechanisms of the action of miraculin, a taste modifying protein” should provide sufficient information for the instructor on miracle fruit and the miraculin protein (10). This reading should facilitate understanding of miraculin as an agonist under acidic conditions and an antagonist under neutral or basic conditions (10). Finally, the instructor can prepare and facilitate active learning by transitioning between small group and whole group discussion, and promoting inquiry-based learning. Instructors who may be incorporating active learning into their classrooms for the first time might find Scientific Teaching by Handelsman, Miller, and Pfund to be a useful resource (13).
Scientific Teaching Themes
Active Learning
The miraculin activity exhibits many of the components of active learning outlined in a 2014 meta-analysis of active-learning studies, as well as in other literature (2, 14, 15). For example, studies have shown that verbal communication with instructors and peers plays a key role in student learning during active assignments (5). Real-time instructor feedback is a key component of this lesson. Students work in small groups, so the activity is social in nature. However, students are additionally given the freedom by the instructor to communicate with peers in other groups. This encourages the articulation and sharing of thought patterns, a key component of active learning. A class discussion of results at the conclusion of the experiments also requires students to communicate their results. Students actively drive their own learning by asking and answering self-designed research questions and forming hypotheses relative to these questions. Students also generate their own data collection mechanisms. Overall this activity epitomizes inquiry-based learning, and therefore takes on an active nature.
Assessment
In order to facilitate maximum student freedom and exploration, we did not formally assess this activity. After the lab, students were asked how they might refine or extend their experimental designs, and in this way they assessed their own learning (Supporting File S5: Miracle Fruit: Laboratory Protocol). They were also given a survey in which they assessed their learning of both experimental concepts and receptor binding (Supporting File S3: Miracle Fruit: Post-Activity Survey). Since one of our primary goals was to maximize students’ abilities to take risks and explore the scientific process, we chose to avoid extensive written assignments. While this activity was not formally assessed through a graded assignment, the instructor assessed students’ initial design proposals before they completed their experiment and provided real-time feedback (Supporting File S5: Miracle Fruit: Laboratory Protocol). The instructor used class discussions to monitor student learning throughout the activity in the absence of a formal assessment.
We recognize that other instructors’ goals may differ and may require a formal assessment. If this is the case, there are many feasible options. Pre-lab questions relating to the Misaka article could be graded as a gauge of what students understood prior to completing the activity (Supporting File S4: Miracle Fruit: Pre-lab Questions). Additionally, if a summative assessment is desired, the instructor could require that student groups formally present their experimental design, results, and limitations to the class, thus assessing quality of design and communication. The instructor has the choice of assigning a formal lab report to be completed by students, either in their groups or individually, outside of class after the activity. The instructor could also require students to keep a detailed account of their scientific question, procedure, and results in a lab notebook format and utilize this as an assessment if they choose.
Inclusive Teaching
Many factors are likely to affect an individual student’s exposure to experimental design inside and outside of the classroom. These include, but are not limited to, socioeconomic status and first-generation status. Instructors of upper-level courses might assume that students have a refined understanding of experimental design by that point in their education. However, depending on the quality of introductory courses that students have experienced, this may not be the case. The same is true with respect to the inequities among first-year students and their high school courses. The miraculin activity does not make assumptions as to a student’s level of experimental design experience. It seeks to correct misconceptions and encourages peer support. Additionally, students are encouraged to share their past experience or inexperience with experimental design. Students are given many opportunities and contexts in which to express their knowledge, questions, and concerns. They can express their knowledge in a written manner via pre-lab questions (Supporting File S4: Miracle Fruit: Pre-lab Questions), the laboratory protocol (Supporting File S5: Miracle Fruit: Laboratory Protocol), and the post-activity reflection survey (Supporting File S3: Miracle Fruit: Exit Survey). They can also share knowledge verbally with the instructor one-on-one or during a class discussion. They express their knowledge to peers both within and outside of their lab groups. Therefore, students can choose to whom, when, and in what format they share what they have learned. The level of student choice exhibited throughout the experimental design process allows students to tailor their learning in a way that makes sense to them, promoting inclusivity by allowing different students to learn and experience the activity in different ways. Particularly, this contributes to inclusivity for students with disabilities because it gives them the opportunity to express information in ways that work for them. For example, choosing to present ideas verbally rather than in writing may help students with certain disabilities. On the other hand, students with disabilities related to interpersonal interactions can choose to express their ideas in writing. In general, active learning increases performance of underrepresented and first-generation students by a larger margin than other students (16). Since this activity contains many elements of active learning, it should therefore contribute to inclusivity.
Additionally, the activity allows students to bring their cultural experiences into the classroom and share them with their classmates. For example, students are encouraged to bring in foods that are culturally relevant to them. This shows students the importance of representative sampling in experimental designs. By giving students freedom, the activity encourages them to challenge dominant narratives in science. For example, students from non-Western backgrounds may identify variables or design flaws that Westernized science might miss. This could generate a class discussion after the activity about the relationship between experimental design, diversity, Westernization, and drawing conclusions.
Lesson Plan
Before Class
Suggested Materials
Table 1 highlights three items that instructors should complete prior to teaching the lesson. Foremost, they need to acquire a sufficient amount of miracle fruit from Richadella dulcifica such that each student can try some. Miracle fruit is available from Ethan's Garden. If the instructor cannot obtain miracle fruit, they can also purchase miracle fruit tablets from Miracle Fruit USA. There are also multiple options for purchasing tablets on Amazon, however we have had success with the brand highlighted above. However, the fruit is superior to the tablets because the tablets can degrade over time, whereas the fruit is shipped fresh. Instructors should acquire one fruit or tablet per student. If tablets are used, the instructor must explain to students the proper way to ingest the tablets so that they experience the maximum effect. The students must not chew or swallow the tablets. They must allow the tablet to dissolve completely on their tongue, a process which should take 3–5 minutes. During this time, they should move the tablet around on their tongue so that miraculin can bind to taste receptors on all parts of the tongue. This information can be found in the instructions that come with the tablets.
Exploring Miracle Fruit: Lesson Plan Timeline
| Activity | Description | Estimated Time | Notes |
|---|---|---|---|
| Preparation for Class | |||
| Acquire Miracle Fruit or Tablets |
Order Miracle Fruit from Ethan's Garden or tablets from Miracle Fruit USA |
Allow sufficient time for shipping, about 3-5 days prior to teaching lesson. |
|
| Outside Reading |
|
10 minutes, 3-7 days prior to teaching lesson |
Pre-lab questions can be found in Supporting File S4. Miracle Fruit - Pre-lab Questions |
| Obtain Food |
|
20-60 minutes, 1-2 days prior to teaching lesson | |
| Class Session | |||
| Presentation Part 1: Receptor Binding |
|
5-10 minutes | PowerPoint Slides can be found in Supporting File S1. Miracle Fruit - Presentation Slides |
| Pre-lab Discussion |
|
5-10 minutes | Pre-lab questions can be found in Supporting File S4. Miracle Fruit - Pre-lab Questions |
| Discussion of Laboratory Protocol |
Allow students to read laboratory protocol and answer questions within their small groups |
10 minutes | Laboratory Protocol can be found in Supporting File S5. Miracle Fruit - Laboratory Protocol |
| Students Design Experiments |
|
40 minutes | Supporting File S2. Miracle Fruit - Probing Questions to Uncover Experimental Design Issues can aid the instructor in critiquing student designs |
| Students Conduct Experiments |
Allow students to carry out their approved experimental procedures |
20-60 minutes | Can overlap with Presentation Part 2 |
| Presentation Part 2: Mechanism of Miraculin |
Present slides 11-17 of “Receptors and Biochemistry” PowerPoint |
20 minutes |
|
| Data Analysis |
Allow students to draw conclusions about their results within their groups |
20 minutes | Can overlap with class discussion |
| Class Discussion of Results |
|
10-30 minutes | Can overlap with data analysis; Supporting File S2. Miracle Fruit - Probing Questions to Uncover Experimental Design Issues may be useful to instructor |
Instructors can provide foods for students to try with the miracle fruit, and they can also ask students to bring in foods that they would like to try with the miracle fruit. The experiments that students design will be centered around the foods that students bring in as well as those provided by the instructor. We provided lemons, limes, tabasco sauce, antacid tablets (e.g., Tums), sugar, artificial sweeteners, cherry tomatoes, cider vinegar, salt and vinegar potato chips, arugula, blue cheese crumbles, dark and milk chocolate chips, sweet and dill pickles, coffee, pomegranate juice, celery, broccoli, blueberries, strawberries, and avocado. We chose these foods due to their variations in pH and taste profiles, factors which many students choose to utilize as the independent variable in their experiments. Lemons, limes, and cherry tomatoes taste very different with miraculin, so their use in the experiment ensures that students experience maximum effect. We emphasize the use of avocado as one of the few alkaline foods, to widen the pH range of the food selection. Instructors should check with their classes a few days before the activity to ensure that no students have food allergies. Due to common allergic issues and lack of significant taste differences, we recommend avoiding nuts entirely. Students may want to bring in favorite foods of theirs that they wish to experiment with and try with miracle fruit. Students can choose to share the foods they bring in with the whole class, or use them only within their group. The instructor-provided foods should be sufficient to conduct an interesting experiment, with student-provided foods serving as supplement. In the interest of equity, no students should be required to bring food to class. If the instructor encourages students to bring in foods, they must make the whole class aware (anonymously) of any allergy issues to avoid any health concerns. Additionally, the activity should be conducted in a food-safe environment (e.g., in a food-safe room, on plastic tablecloths, etc.).
Outside Reading
Three to seven days prior to conducting the activity, the instructor should provide students with access to the article “Molecular mechanisms of the action of miraculin, a taste modifying protein” by Misaka, and the corresponding pre-lab questions (10, Supporting File S4: Miracle Fruit: Pre-lab Questions). Students should read the article carefully and complete these questions prior to the lab (Supporting File S4: Miracle Fruit: Pre-lab Questions). The rest of the lesson is designed to occur during a three-hour lab session.
Lesson Overview
Table 1 outlines the distribution of time across various components of the lesson. Instructors can adapt the lesson to fit shorter or longer class periods, but ideally the experimental design process and physical experimentation occur during a single class period. For example, the instructor could provide students with the background knowledge on receptor binding found in Presentation Part 1 or discuss pre-lab questions during a prior class if necessary. Additionally, discussion of experimental results could occur during a class period following experimentation, but we recommend that it is done during the same class period. Upon arrival to lab, the instructor can place participants into groups of 3 or 4 students. We randomly assigned students to groups in order to maximize inclusivity. Each individual will receive one miracle fruit or tablet upon approval of their experimental design. The instructor should provide all groups with access to the same foods. Students should have their pre-lab questions and Misaka article with them.
Presentation of Background Information
Next, the instructor should present background knowledge on membrane-bound receptor pathways (Supporting File S1: Miracle Fruit: Presentation Slides). This presentation includes diagrams of pathways containing G-proteins, and, very generally, describes the cascade of chemical changes that occurs when a molecule binds to a receptor. The instructor should emphasize the important protein conformational changes. Students should understand the difference between an agonist and antagonist following the presentation. After students have this background knowledge, they should discuss their answers to the pre-lab questions in their groups, followed by a class-wide discussion (Supporting File S4: Miracle Fruit: Pre-lab Questions). This will clarify any remaining misconceptions from the Misaka paper about the known action of miraculin. Next, the instructor should provide the students with the laboratory protocol (Supporting File S5: Miracle Fruit: Laboratory Protocol). This protocol contains three additional questions, which students should ponder on their own and then discuss with their groups. The students should have access to Misaka 2013 while doing this.
Experimental Design Activity
Now, the students can begin to design their experiments in their small groups. Students can choose any experiment that they wish. However, if the instructor wishes, they can use a more guided-inquiry approach and provide the students with a few choices for their central question. The discussions at the beginning of the class should provide them with some ideas (Supporting File S2: Miracle Fruit: Probing Questions to Uncover Experimental Design Issues). Students commonly investigate the effect of miraculin over time. Others explore the differences in the effects of miraculin on foods of different pH and/or sweetness/bitterness. Students have access to the internet to obtain any information about the foods that they wish. The challenge lies in crafting an experiment that quantifies taste perception and is as controlled as possible (see Supporting File S2: Miracle Fruit: Probing Questions to Uncover Experimental Design Issues for important guidance). Because the goal is to expose misunderstandings of experimental design and allow for the correction of those misunderstandings, the instructor should recommend that students describe their initial ideas early (e.g., within 20 minutes) so that they have an additional 20 minutes to modify their approach. The instructor may need to guide students in their understanding of the connections between their hypothesis, their predictions, and their chosen experimental design.
Experimental Design Revisions
Student groups must propose their designs to the instructor. Most likely, the initial experimental design will contain confounds, unbeknownst to the students. Often, students misquantify taste; for example, many students think that sweet and sour are opposites without realizing that some foods simultaneously exhibit both sweet and sour taste profiles. Students may ignore inter-individual differences in taste perception; in this case the instructor should direct them to “calibrate themselves” in some way by tasting foods and placing them on a single numeric scale common to the group. Additionally, initial student designs may fail to account for order effects, differences in timing, or inter-individual differences in sensitivity to miraculin. The instructor points out these flaws to groups (see Supporting File S2: Miracle Fruit: Probing Questions to Uncover Experimental Design Issues). Then the groups revise their designs and approach the instructor again. Frequently, the revised experimental designs are still flawed due to lack of subjects or time. This outcome is acceptable, as long as the students can identify what the flaws are and how they are attempting to address these flaws. Pointing out that time, resources, or personnel are limitations in all scientific ventures can help students appreciate how science is actually conducted in laboratories and field sites throughout the world.
When the instructor feels that students have sufficiently addressed the flaws in their original experimental designs, they can approve the student design. After this, they can instruct student groups to conduct their experiments using the procedures they have written. Students collect data, draw conclusions from the data, and propose refinements or extensions of their experiments. While groups complete experiments, the instructor can begin to give an explanatory lecture about the effect of pH on miraculin (Supporting File S1: Miracle Fruit: Presentation Slides). This information is specifically presented after the experimental design process is complete to give students the opportunity to hypothesize about the mechanisms of miraculin on their own as they design their experiments. This mini-lecture should include diagrams of receptor pathways under neutral and acidic conditions. The instructor may briefly explain protein structure and the concept of pH. Depending on the class’s familiarity with these topics, the instructor can choose the level of depth and amount of time spent on these concepts. Carboxylic acid can be used as a sample molecule with which to show the chemical effect of the loss of a proton. While the instructor may be specifically interested in the molecular modeling of miraculin at different pH levels (17), we suggest focusing the lesson on the more generalizable concepts regarding the relationship between pH and protein confirmation. After this discussion, the instructor may wish to show or draw a new diagram of the receptor pathway, showing differences in shape of binding proteins under acidic and neutral conditions, to emphasize that changes in pH lead to conformational alterations.
Analysis and Discussion
Next, the instructor should give the groups some time to analyze their data and draw conclusions based on their results. This can occur prior to or as a part of a class discussion of experiments and results. Groups can then share their findings and conclusions with the whole class. Most importantly, the class should discuss the common flaws and confounds in their experimental designs and how to minimize these. Students can propose new, improved experimental designs or research questions following this discussion. It is important to emphasize that their experiments are not useless; rather, their data provides information that simply has limits regarding how generalizable it is.
Teaching Discussion
Achievement of Learning Goals and Objectives
During two iterations of this course (two lab sections in 2014, two more in 2015), students were asked to fill out a survey following the completion of the activity. The instructor informed the students that this survey would not affect their grade, and that the instructor would not have access to the completed surveys until after grades were submitted at the end of the semester. The survey included self-assessment questions in which students evaluated their own learning using Likert scales (Supporting File S3: Miracle Fruit: Exit Survey). Students also had the opportunity to provide the instructor with feedback on the effectiveness of the activity. Students were not given a grade for the activity.
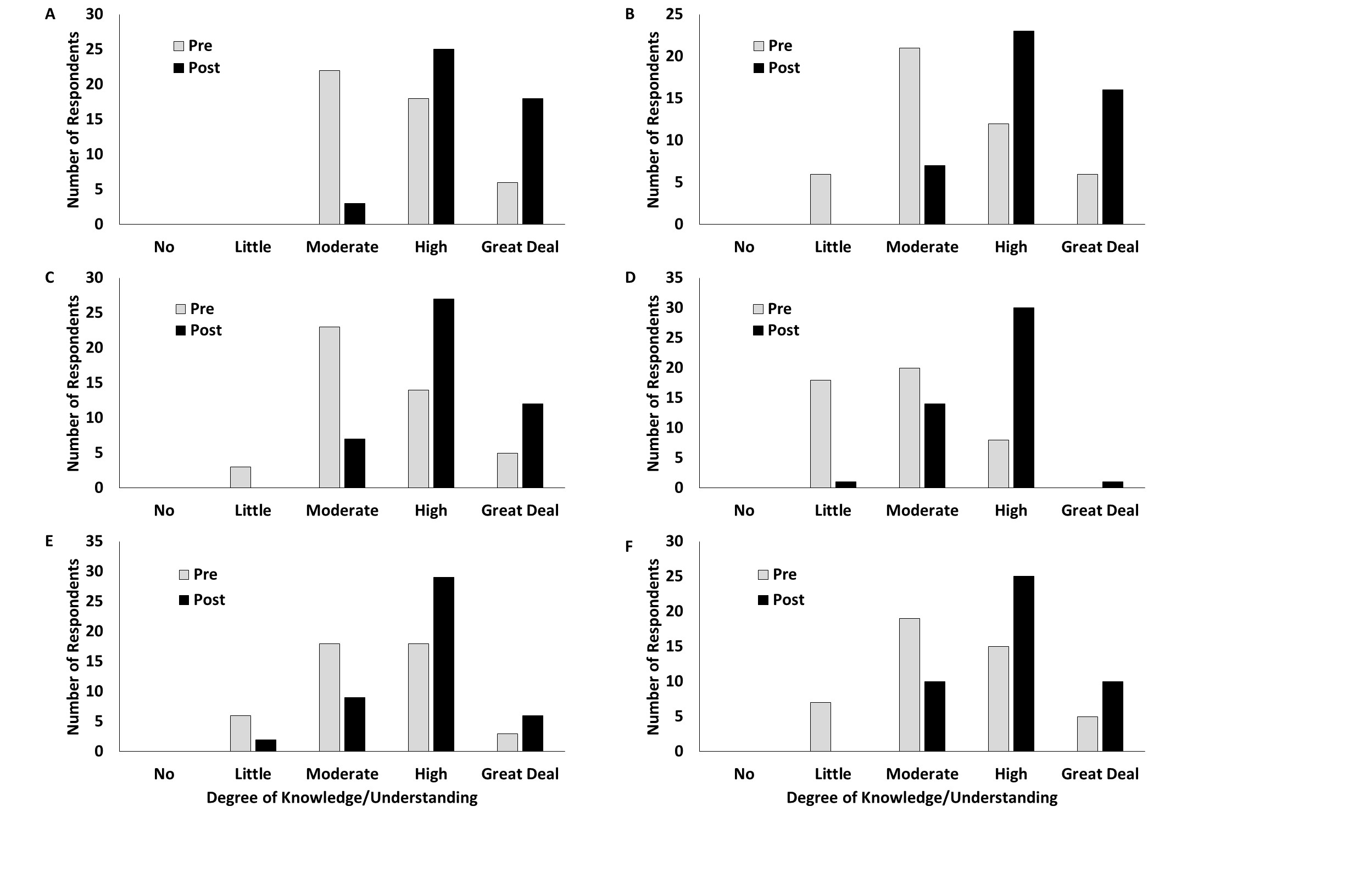
Overall, students reported that the activity increased their knowledge of various aspects of experimental design. Specifically, students reported a statistically significant increase in understanding regarding general rules of how to design an experiment (χ2 = 21.56, d.f. = 2, P < 0.0001, Figure 1), detailed factors of experimental design (χ2 = 20.99, d.f. =3, P = 0.0001, Figure 1), and the ability to address natural phenomena drawing on multiple disciplines (χ2 = 18.53, d.f. = 3, P = 0.0003; Figure 1). Additionally, participants reported increased knowledge of biochemistry-related topics, including taste (χ2 = 30.01, d.f. = 3, P < 0.0001, Figure 1), receptor binding (χ2 = 13.95, d.f. = 3, P = 0.0030, Figure 1), and the dynamic nature of macromolecules (χ2 = 8.56, d.f. = 3, P = 0.0357; Figure 1). However, there were no reported increases in knowledge with respect to pH (χ2 = 3.64, d.f. = 2, P = 0.1622), likely due to an already high degree of understanding prior to the activity. If the activity were utilized in an introductory biology class, however, it is possible that it would play a greater role in increasing student knowledge of pH, because students would enter with less background knowledge on the subject.
Generally, students showed an appreciation for the complexities and challenges involved in designing an objective experiment. One student noted, “This experiment helped me see some of the flaws and shortcomings in many experiments,” suggesting an ability to apply the knowledge gained during the activity to other experimental situations. Multiple students explicitly stated that they had realizations about the importance of nested factors including order effects and inter-individual variation. For example, one student reported in their survey that they “have a better understanding of ways to limit confounds,” while another student said that they “learned about time effects and nesting trials and how that may affect results.”
Students also expressed satisfaction with the opportunity to learn and clarify biochemistry concepts while doing this experiment. When asked specifically what they learned, students frequently cited the relationship between pH and protein shape, as well as taste receptor function. For example, one student stated, “I learned more about how pH affects the likelihood of protonation, which affects the form of a protein, which affects its function.” Another student expressed surprise at their discovery about taste receptor function: “I learned a lot about how taste receptors work and I was surprised to learn that the miraculin permanently binds to the sweet receptors but the receptors are changed every hour or so.” Lastly, students reported enjoyment of the activity, with all students stating that they enjoyed it at some level (Figure 2). Particularly, students reported that the hands-on nature of the activity, the level of independence in the design process, and the variety of foods contributed to their enjoyment of the activity.
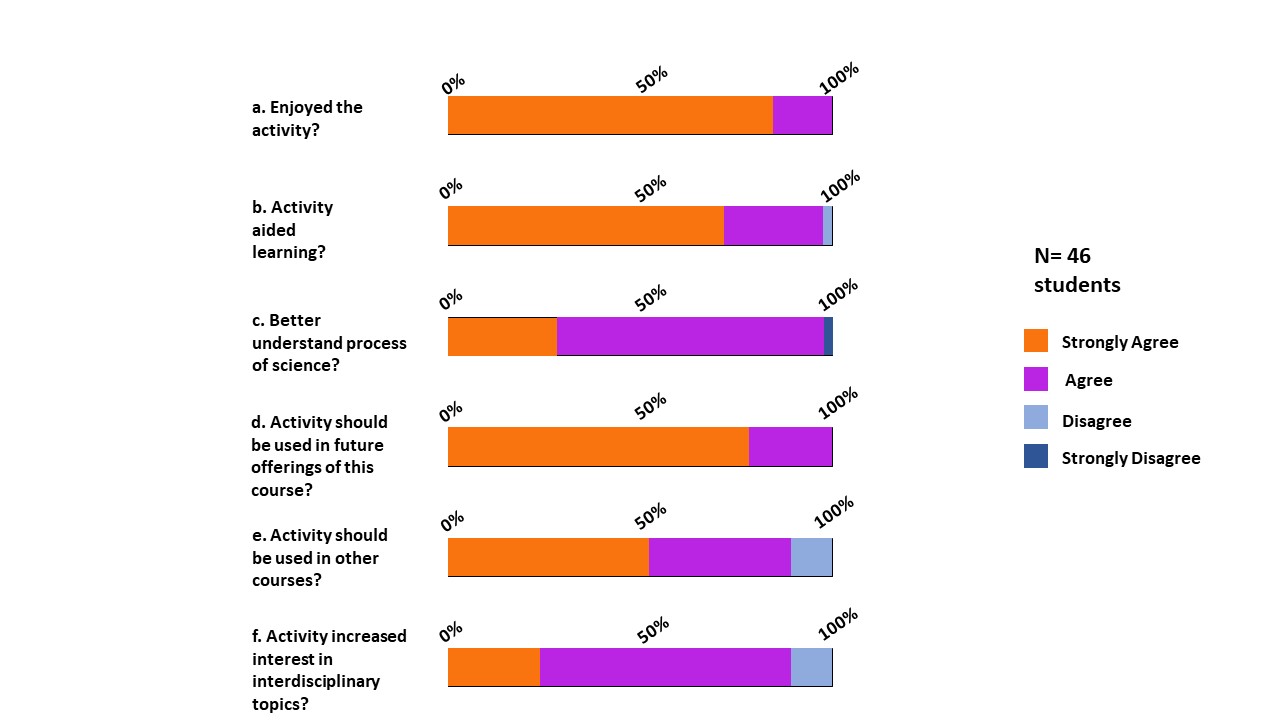
We used this activity as a preparatory measure for a 5-week CURE that occurred later in the semester. This lesson exposes participants to the five dimensions of CUREs: use of scientific practices, discovery, broadly relevant or important work, collaboration, and iteration (7). Because the miraculin activity requires students to follow the whole scientific process, from reviewing prior literature to communicating results, they become more qualified to conduct research in the context of a CURE. Experience with scientific work on a smaller scale may make students more confident when they delve into a CURE, thus enabling them to achieve maximum potential benefit.
Adaptations
This activity can be easily adapted by instructors to suit biochemistry or introductory biology courses. Since it focuses on experimental design, the lesson inherently has value for any science student. Details can be altered to fit different student populations. For example, in an introductory biology course, an instructor might provide more guidance during the review of Misaka 2013, as first-year students likely have less experience analyzing scientific papers. The paper could be analyzed during a class period prior to completion of the activity, or the instructor could assign groups prior to the day of the activity and have students collaborate in their analysis of the paper. Additionally, the instructor might place less emphasis on more advanced concepts such as agonist and antagonist binding, and focus more on the topics covered in a typical introductory course, such as the flow of information in the receptor pathway or the three-dimensional structure of proteins. Whereas our students developed their initial scientific questions independent of instructor suggestions, introductory biology students may require more guidance throughout the experimental design process. In this case, the instructor could provide some general examples of the types of scientific questions that students can explore regarding miracle fruit, and allow the students to choose and develop the specifics of the design. A biochemistry course may delve deeper into the specific pH-dependent conformational changes of the miraculin protein and its effect on the receptor. Overall, this activity exhibits diverse value across disciplines by teaching students how to think like scientists.
SUPPORTING MATERIALS
- S1: Miracle Fruit: Presentation Slides
- S2: Miracle Fruit: Probing Questions to Uncover Experimental Design Issues
- S3: Miracle Fruit: Exit Survey
- S4: Miracle Fruit: Pre-lab Questions
- S5: Miracle Fruit: Laboratory Protocol
Acknowledgments
This project was reviewed and approved by the Institutional Review Board (IRB) at Lafayette College (protocol # AY1415-02), and was supported by the Camille and Henry Dreyfus Foundation (Grant No. TH-18-017).
References
- DebBurman SK. 2002. Learning How Scientists Work: Experiential Research Projects to Promote Cell Biology Learning and Scientific Process Skills. Cell Biol Educ. 1:154-172.
- Bauerle C, DePass A, Lynn D, O’Connor C, Singer S, Withers M, Anderson CW, Donovan S, Drew S, Ebert-May D, Gross L, Hoskins SG, Labov J, Lopatto D, McClatchey W, Varma-Nelson P, Pelaez N, Poston M, Tanner K, Wessner D, White H, Wood W, Wubah D. 2011. Vision and Change in Undergraduate Biology Education: A Call to Action. American Association for the Advancement of Science, Washington, DC.
- Luckie DB, Maleszewski JJ, Loznak SD, Krha M. 2004. Infusion of collaborative inquiry throughout a biology curriculum increases student learning: a four-year study of “Teams and Streams.” Adv Physiol Educ. 287:199-209.
- Myers MJ, Burgess AB. 2003. Inquiry-Based Laboratory Course Improves Students’ Ability to Design Experiments and Interpret Data. Adv Physiol Educ. 27:26-33.
- Modell HI, Michael JA, Adamson T, Horwitz B. 2004. Enhancing active learning in the student laboratory. Adv Physiol Educ. 28:107-111.
- Ryder J, Leach J, Driver R. 1998. Undergraduate Science Students’ Images of Science. J Res Sci Teach. 36(2):201-219.
- Auchincloss LC, Laursen SL, Branchaw JL, Eagan K, Hanauer DI, Lawrie G, McLinn CM, Pelaez N, Rowland S, Towns M, Trautmann NM, Varma-Nelson P, Weston TJ, Dolan EL. 2014. Assessment of Course-Based Undergraduate Research Experiences: A Meeting Report. CBE Life Sci Educ 14:29-40.
- Bangera G, Brownell SE. 2014. Course-Based Undergraduate Research Experiences Can Make Scientific Research More Inclusive. CBE Life Sci Educ 13:602-606.
- Corwin LA, Graham MJ, Dolan EL. 2015. Modeling Course-Based Undergraduate Research Experiences: An Agenda for Future Research and Evaluation. CBE Life Sci Educ 14(1):1-13.
- Misaka T. 2013. Molecular mechanisms of the action of miraculin, a taste modifying protein. Semin Cell Dev Biol. 24:222-225.
- Lipatova O, Campolattaro MM. 2016. The Miracle Fruit: An Undergraduate Laboratory Exercise in Taste Sensation and Perception. J Undergrad Neurosci Educ. 15(1): A56-A60.
- Gillen CM. 2006. Criticism and Interpretation: Teaching the Persuasive Aspects of Research Articles. CBE Life Sci Educ. 5(1).
- Handelsman J, Miller S, Pfund C. 2007. Scientific Teaching, The Wisconsin Program for Scientific Teaching, Roberts & Company, and W.H. Freeman, Madison, WI; Englewood, CO; and New York.
- Freeman S, Eddy SL, McDonough M, Smith MK, Okoroafor N, Jordt H, Wenderoth MP. 2014. Active learning increases student performance in science, engineering, and mathematics. Proc Natl Acad Sci USA. 111(23).
- Labov JB, Reid AH, Yamamoto KR. 2010. Integrated Biology and Undergraduate Science Education: A New Biology Education for the Twenty-First Century? CBE Life Sci Educ. 9:10-16.
- Eddy SL, Hogan KA. 2014. Getting Under the Hood: How and for Whom Does Increasing Course Structure Work? CBE Life Sci Educ. 13:453-468.
- Paladino A, Costantini S, Colonna G, Facchiano AM. 2008. Molecular modelling of miraculin: Structural analyses and functional hypotheses. Biochem Biophys Res Comm. 367:26-32.
Article Files
Login to access supporting documents
Ganser-Exploring Miracle Fruit.pdf(PDF | 462 KB)
S1. Miracle Fruit-Presentation Slides.pptx(PPTX | 1 MB)
S2. Miracle Fruit-Probing Questions to Uncover Experimental Design Issues.docx(DOCX | 15 KB)
S3. Miracle Fruit-Exit Survey.docx(DOCX | 99 KB)
S4. Miracle Fruit-Pre-lab Questions.docx(DOCX | 22 KB)
S5. Miracle Fruit-Laboratory Protocol.docx(DOCX | 23 KB)
- License terms

Comments
Comments
There are no comments on this resource.