Data Analysis Recitation Activities Support Better Understanding in SEA-PHAGES CURE
Editor: Rachelle M. Spell
Published online:
Abstract
Course-based undergraduate research experiences (CUREs) are widely known to improve student learning outcomes in the sciences. Undergraduate students have a particularly difficult time interpreting the scientific data that they generate in these experiences – especially when lacking opportunity and exposure to science processes prior to entering higher education. Therefore, it is vital to structure these research experiences such that students can see maximal gains in their skills. This is especially true in the SEA-PHAGES (Science Education Alliance Phage Hunters Advancing Genomics and Evolutionary Science) lab experience, which focuses on bacteriophages – a subject about which most undergraduate students have limited knowledge. In the SEA-PHAGES lab experience at The Ohio State University, we observed that while students made rapid gains in science process skills over the course of the semester, they still struggled to interpret the data they generated. To address this issue, we designed and implemented a set of five recitation activities to complement the lab experience, termed Recitation Activities to Improve Literacy in Science (RAILS). Using an adapted student assessment of learning gains (SALG) survey, we observed that these activities improved students' perceived ability to interpret their data, and students reported that they experienced significant gains in their data analysis ability as a result of the activities. We hope that other SEA-PHAGES instructors will similarly benefit from utilizing these recitation activities as part of their implementation of the curriculum.
Primary image: Phages on RAILS. A bacteriophage is pictured driving a train engine down a track.
Citation
Newman-Griffis AH, Sypolt E, Sagatelova M, Cubonova L, Danhart E, Kulesza AE. 2020. Data analysis recitation activities support better understanding in SEA-PHAGES CURE. CourseSource. https://doi.org/10.24918/cs.2020.48
Lesson Learning Goals
Following these activities, students will:
- Become more confident in their ability to perform laboratory tasks and problem solve in a laboratory setting
- Connect the lab procedures to their outcomes
- Critically analyze/reflect on the data they generate from their experiments
Lesson Learning Objectives
Overall
Broadly, following these activities, students will be able to:
- Explain the goals, procedures, and outcomes of the experiments they perform
- Analyze and interpret the data they generate
- Apply the methods they use to real-world situations
Direct Isolation
Following this activity, students will be able to:
- Describe how to isolate a novel phage from a soil sample using different protocols
- Compare and contrast direct and enriched isolation protocols
- Critically analyze plaque assay results and explain experimental shortfalls
Three-Phase Streak
Following this activity, students will be able to:
- Describe the procedures associated with isolation and purification of a phage from an environmental sample
- Compare and contrast example three-streak plates to critically analyze varying results
- Rationalize differences in three-streak plates and next step procedures
Serial Dilution
Following this activity, students will be able to:
- Calculate PFU titer
- Explain the meaning of PFU
- Differentiate between the serial dilution and titer experiments
DNA Extraction
Following this activity, students will be able to:
- Detail the contents of phage MTL (medium titer lysate)
- Explain the purpose of adding nuclease and resin to the MTL
- Explain the contents of their sample at various stages of the DNA isolation protocol
- Explain the function of resin in the protocol
- Differentiate between filters and columns
- Read a spectrophotometer spectrum
- Explain the significance of the 260/280 ratio in DNA isolations
- Calculate how much DNA to add to a restriction digest reaction based on its concentration
Restriction Enzymes and Gels
Following this activity, students will be able to:
- Explain how restriction enzymes work.
- Identify restriction enzyme cutting sequences within a DNA fragment.
- Differentiate between the number of cutting sites for a restriction enzyme and the number of fragments it creates.
- Explain why and how gel electrophoresis works
- Analyze a gel by determining whether and how many times a restriction enzyme has cut a fragment of phage DNA
- Create a hypothetical gel based on a given sequence or set of fragments
- Compare their gel to known phage samples based on the number and size of DNA fragments generated by restriction enzyme digestion
Article Context
Course
Article Type
Course Level
Bloom's Cognitive Level
Vision and Change Core Competencies
Class Type
Class Size
Audience
Lesson Length
Pedagogical Approaches
Principles of How People Learn
Assessment Type
INTRODUCTION
Undergraduate research has been shown to be effective in helping students understand the scientific process (1) and increase student participation and retention in the sciences (2), especially those from minority populations (3). Undergraduate research experience is often contingent on faculty and graduate student mentorship. However, there are generally not enough faculty researchers available to provide this beneficial experience to undergraduates, and not all faculty are willing or able to provide students with their own projects. This creates a hypercompetitive environment and excludes many students, especially those in marginalized groups, from participating in research throughout their undergraduate career (4). Course-based undergraduate research experiences (CUREs) provide an opportunity for students to learn laboratory techniques as well as engage in authentic experiences using the scientific process (5,6). By participating in authentic research in the classroom, students demonstrate gains in understanding of science process skills, report more confidence in their ability to do research and greater ownership of their projects, and persist longer in the sciences than their counterparts in traditional "cookbook" labs (5,7,8).
The CURE context provides instructors with an excellent opportunity to support student learning of both science process and data analysis skills through effective curriculum design (9). However, these benefits can come with increased student anxiety about the open-ended nature of lab tasks (10), and CURE teaching assistants (TAs) have reported challenges in helping students understand laboratory protocols, especially those who are not familiar with subject materials covered in the laboratory (11). Further, gains in science process skills do not necessarily yield gains in data interpretation skills (12), and both must be effectively addressed in course design for students to see improvements in their learning. Here, we present a suite of resources designed to support student learning of data analysis and science literacy in a nationwide CURE curriculum.
SEA-PHAGES (Science Education Alliance-Phage Hunters Advancing Genomics and Evolutionary Science; 13) involves a year-long project, which at The Ohio State University is taught as part of undergraduate introductory biology courses with an optional second-semester continuation of the project (https://seaphages.org) and is taught at over 120 institutions nationwide. Throughout this lab experience, students learn and utilize key microbiological techniques such as three-phase streak, serial dilutions, and DNA extraction, in order to isolate and characterize a bacteriophage that can infect one of several soil-dwelling bacterial hosts belonging to the Actinobacter phylum. (https://seaphages.org/hosts/). In the implementation of the SEA-PHAGES curriculum at The Ohio State University (OSU), we observed the act of learning and performing the various complex protocols led to rapid gains in science process skills for the students. Within one semester, students become familiar with microbiological and molecular biological techniques such as pipetting, plating, aseptic technique, and identifying the plaques created by phages.
While students were able to make observations about the data they were generating, they lacked the ability to interpret those data. For instance, while students could easily identify and count how many plaques there were on a petri dish, they struggled to draw conclusions about what the numbers and types of plaques indicated about the activity of the phage. This disconnect was reflected in the way that students discussed their data, their ability to troubleshoot, and ultimately in the quality of their final research papers, the capstone project of the OSU SEA-PHAGES implementation. The issue was further exacerbated by the knowledge gap in TAs' ability to help students interpret their data, especially those who are non-experts in the field of virology and microbiology.
To address these issues, we created a centralized set of resources to help students conceptualize their experiments and think critically about the data they generate, and to support instructors in their teaching of data analysis skills. Based on these overarching goals, along with specific learning outcomes that are detailed in each activity, we used backwards design (14,15) to create a set of five recitation activities that could be implemented by instructors during the lab period. These activities, which we called Recitation Activities to Improve Literacy in Science (RAILS), leverage the benefits of group-based problem solving to improve students' critical thinking skills (16) and are designed to complement SEA-PHAGES laboratory procedures. To increase the transparency and inclusivity of the RAILS, we incorporated several elements recommended by the TILT (Transparency in Teaching and Learning) Higher Ed project (17), including transparent learning objectives and instructions. The RAILS were implemented for two semesters and data were collected on their efficacy using the student assessment of learning gains (SALG) survey (18; Supporting File S11), in which students reported a satisfactory to high gain in their learning as a result of completing the RAILS. To our knowledge, this is a unique approach to supporting student outcomes in CURE labs. Here we present these activities (Supporting Files S1-10) and detail our recommendations for their implementation and adaptation to serve the needs of the larger SEA-PHAGES community.
Intended Audience
These activities were designed for first- and second- year STEM majors enrolled in the SEA-PHAGES lab experience. While some diversity exists in the scale and method of SEA-PHAGES curriculum implementation at different institutions, we believe that the RAILS have the potential to be widely applicable to a diversity of CUREs.
At OSU, the SEA-PHAGES lab curriculum is implemented as the laboratory portion of a high-enrollment introductory Biology for STEM majors. The lecture portion of the course is taught by the instructor of record, and the lab portion is taught by teaching associates, who are either graduate students or contract lecturers. Generally, eight sections of the SEA-PHAGES lab comprising 20-24 students are taught each semester. The lab portion meets weekly for 2 hours and 45 minutes and is intended to include time for recitation, administration, quizzing, and lecture exam review, as well as the laboratory activity itself. As many SEA-PHAGES laboratory procedures can take all of the allotted lab time, these additional elements are often completed during downtime within the lab protocols. The RAILS activities were designed to take advantage of these free periods, with a completion time of about 45 minutes each.
As detailed in the adaptations section, while the RAILS activities were designed to parallel the schedule of the SEA-PHAGES lab at OSU, they could easily be reordered and/or adapted to suit alternative implementations, including different host bacteria. As a result of our focus on transparency and inquiry-based learning when designing the RAILS, the activities also have the potential to be used in an online or hybrid implementation of SEA-PHAGES, where they could be used as preparatory activities for the week's lab or to complement video demonstrations of protocols. They further have the potential to be used in non-SEA-PHAGES labs which use the same techniques.
Required Learning Time
The RAILS activities require an average of 45 minutes of class for facilitation. In our implementation of the SEA-PHAGES curriculum, students meet weekly for 2 hours and 45 minutes with an instructor, and we generally found that it is possible for the RAILS activity to be used in tandem with the lab. Implementation during the lab should be considered active time for the instructor, as circulating and guiding students while they complete the activity significantly helps students' understanding. We recommend that the RAILS activities be completed prior to the day's experiment, either during the related lab or the week prior, as several were designed with inquiry-based learning in mind. Further, as the RAILS are designed as instructor-guided group-based problem solving activities, use of the activities as homework assignments is not recommended unless highly scaffolded by the instructor in an online or hybrid context.
Prerequisite Student Knowledge
While prerequisite knowledge of key basic biology concepts, such as macromolecules, virology, and bacterial life cycles would be beneficial, it is not a requirement, as the activities are designed to be inquiry-based. Depending on collective student background knowledge, the instructor may choose to provide supplementary information that will allow students to progress through the activity. Supplementary information could include YouTube video series like the Amoeba Sisters, textbook chapters on virology, and/or introductory information contained in the open-source SEA-PHAGES lab manual (https://seaphagesphagediscoveryguide.helpdocsonline.com/2-0-toc). We also recommend that instructors allow students to utilize as many resources as possible, including their lab manuals and reputable internet sources.
Prerequisite Teacher Knowledge
To facilitate the RAILS activities, instructors should have a basic understanding of bacteriophage biology, molecular biology, and microbiology laboratory technique, including DNA extraction, restriction enzyme digest, 3-phase streak, direct/enriched isolation, titer calculation, and aseptic technique. While familiarizing yourself with the lab manual protocols can be helpful, prior understanding of the overall biological concepts is valuable (https://seaphagesphagediscoveryguide.helpdocsonline.com/2-0-toc).
SCIENTIFIC TEACHING THEMES
Active Learning
Active learning is a set of pedagogical techniques that encourages active student engagement in learning, as opposed to passive modes of instruction such as traditional lectures. Studies have shown that active learning leads to improved student outcomes (19). In this set of activities, we have chosen to make use of the group-based problem solving technique to assist in instruction. Activities will be completed in groups of 2-4 students with the instructor circulating and providing guidance and feedback throughout. By using a group-based problem solving strategy and assigning credit based on group performance, these activities leverage the effects of both task and reward interdependence to improve participation (20).
Assessment
These activities can be adapted to multiple assessment strategies. In the context of the activities, we assess student learning based on their answers to the questions, in the reflection section of the worksheet, and in the class discussion following worksheet completion. Depending on the preferences of the instructor, students can be given credit for their answers based on either completion or accuracy. However, we would recommend that accuracy-based grading be implemented only in a highly-supported in-person learning setting because the questions in these activities are designed to require high-level thinking, and students may become discouraged if they are required to provide accurate answers in the absence of instructor support (21). In either case, based on our own experience, we recommend detailed feedback from the instructor to each individual group and large group discussion following activity completion. To facilitate assessment, we have provided guidance and detailed commentary for each question on each RAILS activity. Further student progress towards the learning goals can then take the form of additional assignments or assessments, such as quizzes, exams, or capstone projects like a research paper.
Inclusive Teaching
Each activity works to address inclusivity using both a group-based problem solving strategy and activity transparency. Improving student confidence and ability to interpret data is a primary goal of the RAILS activities. It has been shown that both group-based problem solving and increased transparency have an outsized, positive impact on underserved students' academic performance, confidence, and sense of belonging (17). Studies have shown that diverse groups (i.e. across racial, gender, area of study, class year lines) are more productive than homogenous groups (22). We recommend that instructors group students to maximize diversity within the groups, using demographic information or surveys as sources of information. The instructor should move through the classroom and guide students as issues arise and encourage group collaboration. In an effort to increase the transparency of the RAILS activities, we have elected to incorporate some of the recommendations of (Transparency in Teaching and Learning) TILT Higher Ed (17). For each activity, we have provided both a) the purpose, and b) skills and knowledge gained. Additionally, some difficult, open-ended questions have been clarified by adding language that describes successful answers. We strongly recommend that in introducing the activity, the instructor highlights the purpose statement and learning objectives, with the goal of bringing them to the attention of the students. By doing so, students who might not otherwise understand instructor expectations as a result of an opportunity gap will be more equipped to meet the desired learning outcomes.
LESSON PLAN
Overview
The RAILS are aimed to give students a deeper understanding of the data that they are collecting and how they will analyze these data after they are collected. By guiding them through understanding the goals and outcomes of each protocol, our goal is to encourage students to think critically about the protocols, which will allow them to more easily troubleshoot, navigate the data, and perform the experiments with higher proficiency.
Pre-Class Preparation
The open-source SEA-PHAGES laboratory manual is used throughout all experiments and is a wonderful reference for the RAILS. (https://seaphagesphagediscoveryguide.helpdocsonline.com/2-0-toc).
It is recommended the instructor first complete the worksheets on their own and review the supplemental material prior to handing out the worksheets to students. The majority of the RAILS are designed to be implemented as printed worksheets, with one copy per person or per group, depending on the preference and budgetary requirements of the instructor. Each activity could also be adapted electronically if both the instructor and students have easy access to devices such as tablets.
Additionally, the activities can be modified by providing students with mini dry erase boards to supplement the worksheet. If you choose to do this, make sure to have markers, erasers, and boards ready. This strategy has the added benefit of giving students opportunities to discuss with peers and, if desired, present their whiteboards to the class for further discussion. Students may also need to bring their laboratory notebook to class, depending on instructor preference.
Opening with a Discussion
Most, if not all, of these recitation activities work well with both an opening and closing discussion. The opening discussion typically consists of asking students which types of methods they will be performing that day in the SEA-PHAGES laboratory and for any questions they may have regarding the instructions. As mentioned above, this is a good time to highlight the purpose and learning outcomes of the activity. These recitation activities are completed prior to performing the lab experiments so that the students can discuss more in-depth details about the reasoning behind why they are using these methods, answer critical thinking questions, and consider better avenues to collect data and record it in their notebook.
Worksheet - Progressing Through the Activity
The worksheets may be completed in pairs or small groups, either for correctness or completion points. The instructor should point students at specific resources if they get stuck, including the lab manual, reputable internet sources, and other students in the class. If utilized in an in-person class setting, we recommend that the instructor circulate throughout the room while students complete the activity, providing guidance and answering questions.
Closing the Activity with a Discussion - how can you interpret the results?
After the worksheets are completed by the students, we recommend follow-up discussions where the instructor prompts students to think critically about the activity and protocol. Common student pitfalls and knowledge gaps are detailed in the facilitation guide for each activity (Supporting File S2. SEA-PHAGES Recitation Activities - Direct Isolation Facilitation Guide, Supporting File S4. SEA-PHAGES Recitation Activities - Three Phase Streak Facilitation Guide, Supporting File S6. SEA-PHAGES Recitation Activities - Serial Dilution Facilitation Guide, Supporting File S8. SEA-PHAGES Recitation Activities - DNA Extraction Facilitation Guide, Supporting File S10. SEA-PHAGES Recitation Activities - Facilitation Guide) and identifying where students might have initially gone wrong with specific questions can often spark productive conversation. We also recommend that instructors make note of which questions students struggle with while they complete the activity and spend extra time on those questions in the discussion, for instance by polling students for their initial answers. This discussion should also provide students with the correct answers to the questions, so that they can assess their own performance and use the activities as study tools. Further assessment of student learning can take the form of lab quizzes and exams.
Outline
The following schedule has been designed for the semester schedule at OSU and may need to be adapted to accommodate varying course schedules and SEA-PHAGES implementations.
Activity 1: Direct Isolation
This worksheet would be presented in the same week/day as direct isolation, ideally during the 2 hour incubation period inherent in the protocol. This extended time period gives the students time to explore the lab manual and learn the basic phages knowledge that they will need for the rest of the semester.
- Supporting File S1. SEA-PHAGES Recitation Activities - Direct Isolation Student Worksheet
- Supporting File S2. SEA-PHAGES Recitation Activities - Direct Isolation Facilitation Guide
Activity 2: Three-Phase Streak
This worksheet was designed to identify main complications that could arise when doing a Three-Phase Streak. It focuses on different results the students could obtain from the procedure and how to interpret the results that they record in their notebook as well as in any capstone project at the end of the course. It should ideally be implemented on the same day as the lab, prior to students completing the lab procedure.
- Supporting File S3. SEA-PHAGES Recitation Activities - Three-Phase Streak Student Worksheet
- Supporting File S4. SEA-PHAGES Recitation Activities - Three Phase Streak Facilitation Guide
Activity 3: Serial Dilution
The most pervasive complication we have seen with students' data interpretation is understanding the difference between the two serial dilutions we perform: one to create a webbed plate that will become MTL (medium titer lysate), and one to assess phage activity (plaque-forming units (PFU)) in the titer assay. This activity is designed to guide students' understanding of the difference between the two and introduce them to titer calculation and the meaning of PFU. This activity should be implemented prior to titer calculation. At OSU, titer is calculated on the same day as the DNA Isolation procedure, so the serial dilution activity is implemented the week prior while students are performing the serial dilutions.
- Supporting File S5. SEA-PHAGES Recitation Activities - Serial Dilution Student Worksheet
- Supporting File S6. SEA-PHAGES Recitation Activities - Serial Dilution Facilitation Guide
Activity 4: DNA Isolation
Students will be isolating DNA in this laboratory, but it is a fast-paced and time-consuming laboratory in which students need more time to dissect the moving parts. Student understanding of different enzymes and components of the protocol is essential for interpretation of data, such as DNA concentration and gel electrophoresis. This activity should ideally be implemented the week prior to DNA isolation, unless considerably more than 3 hours is allotted for DNA isolation.
- Supporting File S7. SEA-PHAGES Recitation Activities - DNA Extraction Student Worksheet
- Supporting File S8. SEA-PHAGES Recitation Activities - DNA Extraction Facilitation Guide
Activity 5: Restriction Enzymes and Gels
Depending on how the SEA-PHAGES curriculum is implemented, students can encounter several challenges in analyzing the results of their restriction enzyme digests. At OSU, these problems are two-fold: first, while students perform their own restriction enzyme digests with isolated phage DNA samples, preparatory staff perform the gel electrophoresis and disseminate results to students. Second, no class time is allotted to help students analyze the resultant gels. This activity is designed to fill in these gaps in instruction by helping students conceptualize how restriction enzyme-digested DNA fragments run on a gel and then how to analyze that gel. This activity should ideally be implemented the same week/day as the restriction enzyme digest protocol, either prior to the protocol or during an incubation. In this case especially, either participation-based or rapid grading during the lab period are recommended, which would allow the students to use the graded activity to help them with their independent gel analysis.
- Supporting File S9. SEA-PHAGES Recitation Activities - Restriction Enzymes and Gels Student Worksheet
- Supporting File S10. SEA-PHAGES Recitation Activities - Restriction Enzymes and Gels Facilitation Guide
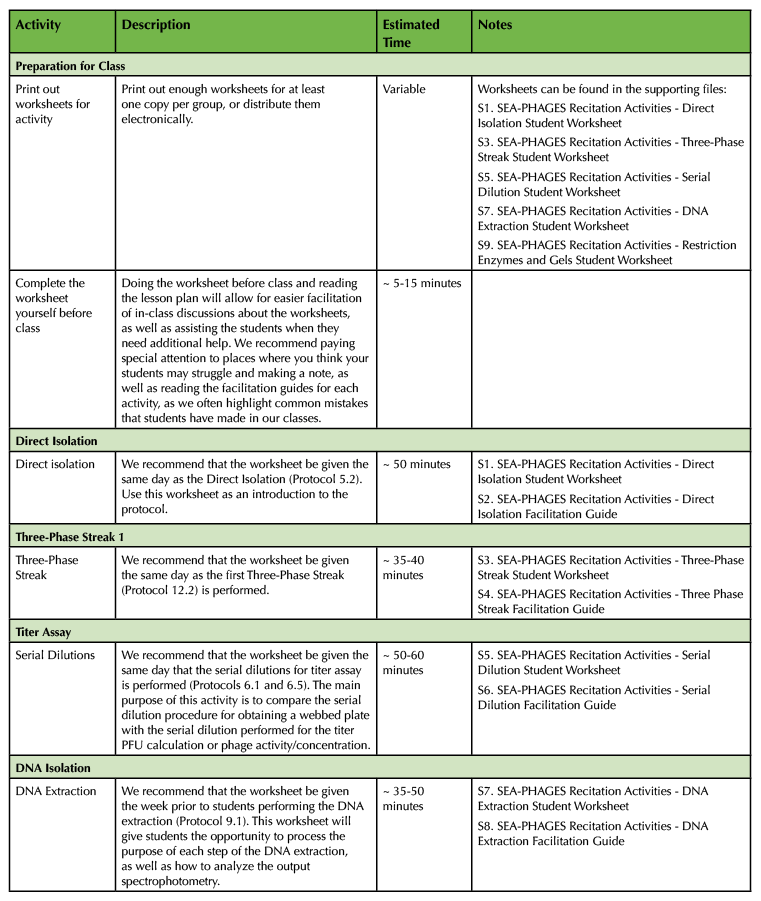
Table 1. RAILS Teaching Timeline. This table indicates the amount of teaching and preparation time needed for each activity, and lists which supporting files correspond to which activity. Timelines for individual activities are provided in the RAILS Worksheet Key, Facilitation Guide, and Timeline files.
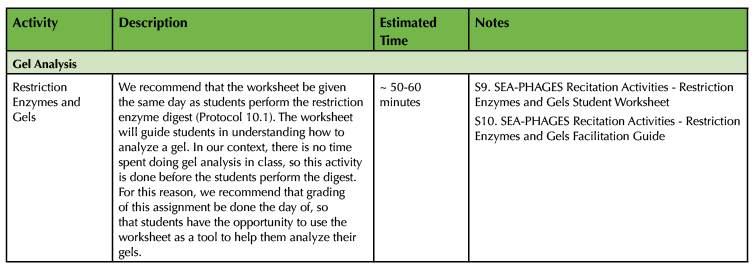
Table 1. RAILS Teaching Timeline (continued). This table indicates the amount of teaching and preparation time needed for each activity, and lists which supporting files correspond to which activity. Timelines for individual activities are provided in the RAILS Worksheet Key, Facilitation Guide, and Timeline files.
TEACHING DISCUSSION
Lesson Effectiveness
We observed in the context of the SEA-PHAGES lab experience at OSU that students were struggling to understand the experiments they were performing and the data they were generating. Based on our observational data from SEA-PHAGES instructors at OSU, we found that the RAILS activities were associated with improved student performance in these two key areas. Qualitative analysis of students' answers on assessments, such as the lab final exam, indicated that student answers demonstrated a higher level of understanding compared to prior semesters. This was especially the case for data interpretation and understanding of the purpose of the experiments they were performing. Additionally, we noticed differences in student ability to recall course-relevant material and troubleshoot what was occurring in their own petri dishes.
To further assess the effectiveness of the RAILS activities, we collected student self-assessment data on whether the RAILS increased student understanding of the lab procedures and helped them interpret their data. Student data were collected using the Student Assessment of Learning Gains (SALG) survey (18), which has been used for course assessment in prior semesters. The SALG survey is designed to summarize learning gains from the student's perspective that result from a given teaching method, avoiding biases that may exist around the course context or instructor. A total of 245 students 18 years and older consented to participate in the study in Autumn 2019 and Spring 2020. The study was determined exempt by the OSU internal review board (IRB#2019E1104).
Five questions were added to the SALG survey to explicitly assess students' perception of their own learning as a result of the RAILS activities that align with the learning goals and outcomes of the lesson (Supporting File S11). These questions asked students to rate their gain in confidence in understanding of experimental goals ("Goals," Figure 1), ability to interpret experimental data ("Data," Figure 1) overall ability as a research scientist ("Ability," Figure 1), executing the laboratory procedure ("Execution," Figure 1), and ability to identify and solve problems encountered during the laboratory procedure ("Troubleshooting," Figure 1), as a result of completing the RAILS activities. Full text of the questions that were added to the SALG are provided in Supporting File S11. SALG Questions Added to SALG Survey to Assess the RAILS Activities. Similar to standard SALG questions, these utilized a five-point Likert-type scale, where students were asked to rate their confidence gains on a scale of 1 (No Gain) to 5 (Great Gain). As reported in E. Seymour et al. (18), scores of 1 or 2 are considered low, scores of 3 or 4 are considered satisfactory to high, and a score of 5 would indicate a strong impact. SALG data were analyzed by averaging student responses (n = 245) on the 5-point scale for each question. Students on average rated their gains as between moderate (3) and good (4), with mean values consistently between 3.6 and 4 (Figure 1). This indicates a satisfactory to high gain in student learning as a result of the RAILS activities (18).
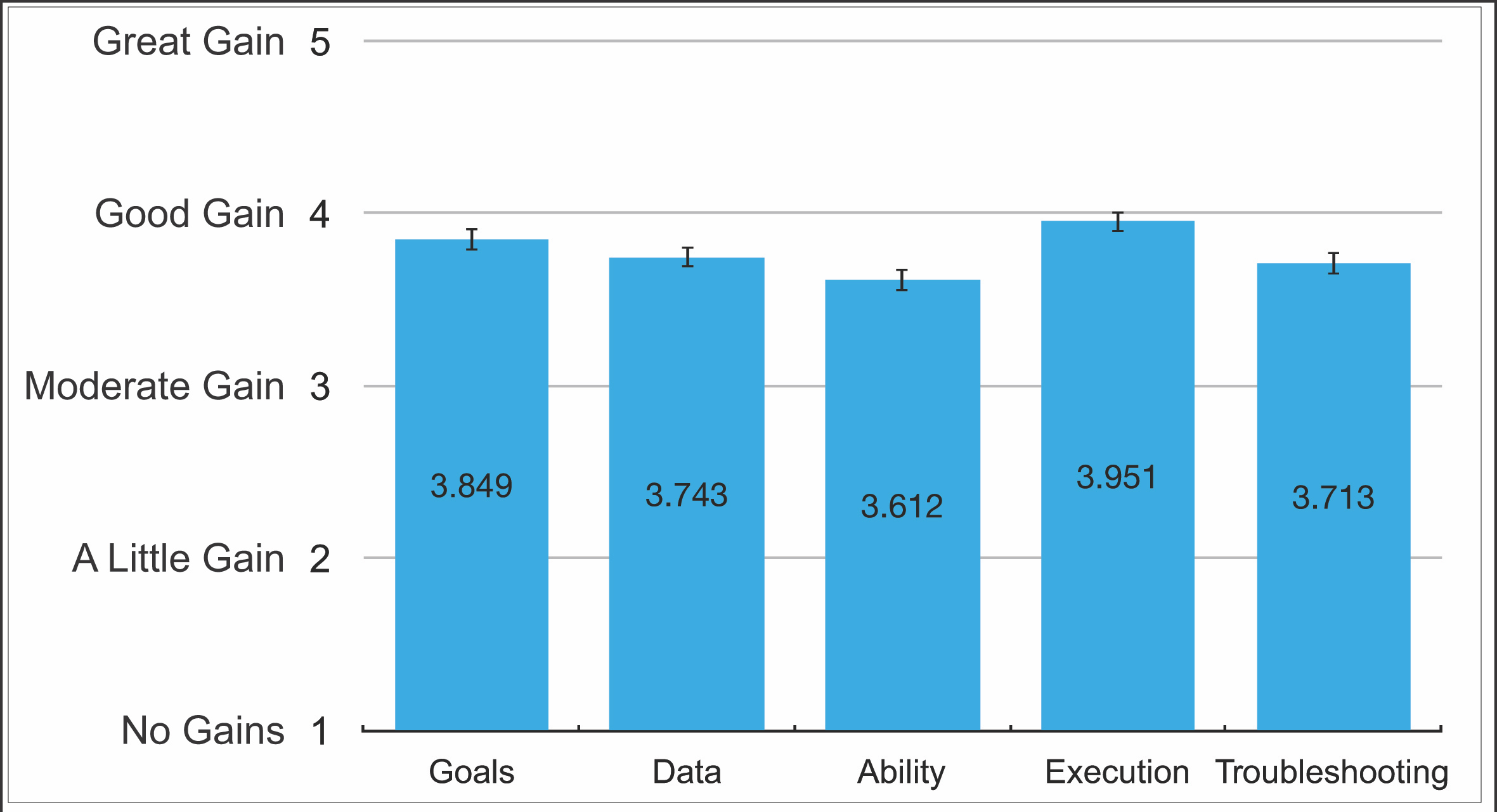
Figure 1. Quantified SALG data from students who utilized the RAILS activities. Students were asked to rate their gains using a 5-point likert scale in five key areas as a result of the RAILS: understanding of experimental goals (Goals), ability to interpret data (Data), confidence in overall ability (Ability), confidence in executing procedures (Execution) and ability to identify and solve problems in the lab procedure (Troubleshooting). Meanings of likert-scale numbers are indicated on the left. Student responses were averaged over all respondents. Bars are mean score, with the exact mean indicated within the bar. N=245. Error bars= standard error.
As students consented to data collection from the entire SALG survey, we were also able to collect student responses to open-ended, qualitative questions. In particular, in response to the question "Please comment on how the class activities helped your learning," several students in both Fall 2019 and Spring 2020 cited the RAILS as particularly helpful to their learning, saying: "The class activities [RAILS] provided in the SEAPHAGES [sic] lab helped my learning significantly. The worksheets given helped in order to review and break up information in a way that was easier to understand and digest," "The RAILS were very useful. It was a nice introduction to the things that we were going to do in the lab," and "The In Class Activities [in lecture] and Recitation Worksheets [RAILS] were always helpful in learning to apply the information I'd learned." These qualitative data are consistent with our quantitative dataset and indicate that overall, students benefited from the RAILS.
Adaptations
These activities were primarily created to work in conjunction with the SEA-PHAGES laboratory (23). Thus, SEA-PHAGES lab instructors can feel free to modify and reorder these activities to fit their own schedule and host bacteria and exclude any data interpretation activities that do not coincide with the methods they are using. Courses that are not using the SEA-PHAGES curriculum can use any of these activities for methods as they see fit. For instance, gel electrophoresis is often used in many classes and thus the activity S9. SEA-PHAGES Recitation Activities - Restriction Enzymes and Gels Student Worksheet could be implemented and modified to work with a number of classes. The same is true for the other activities, which other biology/microbiology courses could use in their own laboratories, edited and refined for each course's needs.
Adapting to Remote Learning
In Spring 2020, the Gel Analysis RAILS activity was implemented in an online instructional setting following student isolation mandated by COVID-19. In this context, the RAILS activity was assigned as homework, with students completing the activity prior to the weekly "lab" meeting over Zoom. The activity was then reviewed during the Zoom meeting, with students asking questions and instructors reviewing the correct answers for the activity. We found it possible to complete this exercise within a 50 minute time frame, depending on the instructor and class population. We feel that all the RAILS activities could be used in an online or hybrid context, especially as an inquiry-based introduction to the lab topic to be completed at home, to minimize the amount of time that students need to spend in a close-proximity lab setting. However, as the activities were designed to be completed in groups, they could prove difficult for students working on their own. In this context, additional scaffolding in the form of instructor-created introductory materials, or use of publicly available video or text content, could be helpful in supporting students' learning.
Another possible adaptation of the activities to an online setting would be to have students complete the activities in breakout groups during a synchronous online "lab." We would expect that this would require additional time, with the activity likely taking 60-80 minutes to complete, because of the additional difficulty of communication introduced by the technology both among students and between instructor and students.
SUPPORTING MATERIALS
- Supporting File S1. SEA-PHAGES Recitation Activities - Direct Isolation Student Worksheet
- Supporting File S2. SEA-PHAGES Recitation Activities - Direct Isolation Facilitation Guide
- Supporting File S3. SEA-PHAGES Recitation Activities - Three-Phase Streak Student Worksheet
- Supporting File S4. SEA-PHAGES Recitation Activities - Three Phase Streak Facilitation Guide
- Supporting File S5. SEA-PHAGES Recitation Activities - Serial Dilution Student Worksheet
- Supporting File S6. SEA-PHAGES Recitation Activities - Serial Dilution Facilitation Guide
- Supporting File S7. SEA-PHAGES Recitation Activities - DNA Extraction Student Worksheet
- Supporting File S8. SEA-PHAGES Recitation Activities - DNA Extraction Facilitation Guide
- Supporting File S9. SEA-PHAGES Recitation Activities - Restriction Enzymes and Gels Student Worksheet
- Supporting File S10. SEA-PHAGES Recitation Activities - Restriction Enzymes and Gels Facilitation Guide
- Supporting File S11. SEA-PHAGES Recitation Activities - Questions Added to SALG Survey to Assess the RAILS Activities
ACKNOWLEDGMENTS
The authors would like to thank Dr. Maggie Wetzel, Dr. Sarah Ball, Dr. David Sovic, Dr. Viknesh Sivanathan, Dr. Sara Faust, and Dr. Rosa Rodríguez Peña for their manuscript feedback and productive discussions. Dr. Stephen Pearson, Ally Langley, Denisha Parker, and Kendall King contributed to RAILS development and experimental design. Dr. Erica Szeyller, Dr. Judy Ridgway, and Dr. Caroline Breitenberger provided key institutional, professional, and emotional support. Thanks to all the students, family, and friends who contributed feedback and support.
References
- AAAS. 2011. Vision and change in undergraduate biology education: A call to action. American Association for the Advancement of Science, Washington, DC.
- Seymour E, Hunter AB, Laursen SL, Deantoni T. 2004. Establishing the benefits of research experiences for undergraduates in the sciences: First findings from a three-year study. Sci. Educ. 88:493-534. http://dx.doi.org/10.1002/sce.10131
- Villarejo M, Barlow AEL, Kogan D, Veazey BD, Sweeney JK. 2008. Encouraging Minority Undergraduates to Choose Science Careers: Career Paths Survey Results. CBE Life Sci. Educ. 7:394-409. http://dx.doi.org/10.1187/cbe.08-04-0018
- Bangera G, Brownell SE. 2014. Course-Based Undergraduate Research Experiences Can Make Scientific Research More Inclusive. CBE Life Sci. Educ. 13:602-606. http://dx.doi.org/10.1187/cbe.14-06-0099
- Auchincloss LC, Laursen SL, Branchaw JL, Eagan K, Graham M, Hanauer DI, Lawrie G, McLinn CM, Pelaez N, Rowland S, Towns M, Trautmann NM, Varma-Nelson P, Weston TJ, Dolan EL. 2014. Assessment of Course-Based Undergraduate Research Experiences: A Meeting Report. CBE Life Sci. Educ. 13:29-40. http://dx.doi.org/10.1187/cbe.14-01-0004
- Linn MC, Palmer E, Baranger A, Gerard E, Stone E. 2015. Undergraduate research experiences: impacts and opportunities. Science 347. http://dx.doi.org/10.1126/science.1261757
- Hanauer DI, Dolan EL. 2014. The project ownership survey: measuring differences in scientific inquiry experiences. CBE Life Sci. Educ. 13:149-158. http://dx.doi.org/10.1187/cbe.13-06-0123
- Hanauer DI, Graham MJ, Betancur L, Bobrownicki A, Cresawn SG, Garlena RA, Jacobs-Sera D, Kaufmann N, Pope WH, Russell DA, Jacobs WR, Sivanathan V, Asai DJ, Hatfull GF. 2017. An inclusive Research Education Community (iREC): Impact of the SEA-PHAGES program on research outcomes and student learning. Proc. Natl. Acad. Sci. 114:13531-13536. http://dx.doi.org/10.1073/pnas.1718188115
- Brownell SE, Hekmat-Scafe DS, Singla V, Seawell PC, Imam JFC, Eddy SL, Stearns T, Cyert MS. 2015. A High-Enrollment Course-Based Undergraduate Research Experience Improves Student Conceptions of Scientific Thinking and Ability to Interpret Data. CBE Life Sci. Educ. 14:1-14. http://dx.doi.org/10.1187/cbe.14-05-0092
- Deters KM. 2005. Student opinions regarding inquiry-based labs. J. Chem. Educ. 82:1178-1180. http://dx.doi.org/10.1021/ed082p1178
- Heim AB, Holt EA. 2019. Benefits and Challenges of Instructing Introductory Biology Course-Based Undergraduate Research Experiences (CUREs) as Perceived by Graduate Teaching Assistants. CBE Life Sci. Educ. 18:1-12. http://dx.doi.org/10.1187/cbe.18-09-0193
- Nixon RS, Godfrey TJ, Mayhew NT, Wiegert CC. 2016. Undergraduate student construction and interpretation of graphs in physics lab activities. Phys. Rev. Phys. Educ. Res. 12. http://dx.doi.org/10.1103/PhysRevPhysEducRes.12.010104
- Jordan TC, Burnett SH, Carson S, Caruso SM, Clase K, DeJong RJ, Dennehy JJ, Denver DR, Dunbar D, Elgin SC, Findley AM, Gissendanner CR, Golebiewska UP, Guild N, Hartzog GA, Grillo WH, Hollowell GP, Hughes LE, Johnson A, King RA, Lewis LO, Li W, Rosenzweig F, Rubin MR, Saha MS, Sandoz J, Shaffer CD, Taylor B, Temple L, Vazquez E, Ware VC, Barker LP, Bradley KW, Jacobs-Sera D, Pope WH, Russell DA, Cresawn SG, Lopatto D, Bailey CP, Hatfull GF. 2014. A broadly implementable research course in phage discovery and genomics for first-year undergraduate students. mBio 5:1-8. http://dx.doi.org/10.1128/mBio.01051-13
- Fink LD. 2013. Creating significant learning experiences an integrated approach to designing college courses. Jossey-Bass, San Francisco.
- Wiggins GP, McTighe J. 2005. Understanding by design. Expanded 2nd ed. Association for Supervision and Curriculum Development, Alexandria, VA.
- Kim K, Sharma P, Land SM, Furlong KP. 2012. Effects of Active Learning on Enhancing Student Critical thinking in an Undergraduate General Science Course. Innov. High. Educ. 38:223-235. https://doi.org/10.1007/s10755-012-9236-x
- Winkelmes M-A, Bernacki M, Butler J, Zochowski M, Golanics J, Weavil KH. 2016. A Teaching Intervention that Increases Underserved College Students' Success. Peer Rev. 18. https://www.aacu.org/peerreview/2016/winter-spring/Winkelmes
- Seymour E, Weise D, Hunter A, Daffinrud S. 2000. Creating a Better Mousetrap: On-line Student Assessment of their Learning Gains, abstr National Meeting of the American Chemical Society, San Francisco, CA, March 27, 2000. https://www.salgsite.org/docs/SALGPaperPresentationAtACS.pdf.
- Freeman S, Eddy SL, McDonough M, Smith MK, Okoroafor N, Jordt H, Wenderoth MP. 2014. Active learning increases student performance in science, engineering, and mathematics. Proc Natl Acad Sci U S A 111:8410-5. 10.1073/pnas.1319030111
- Wilson KJ, Brickman P, Brame CJ. 2018. Group Work. CBE Life Sci. Educ. 17:1-5. http://dx.doi.org/10.1187/cbe.17-12-0258
- Schinske J, Tanner K. 2014. Teaching More by Grading Less (or Differently). CBE Life Sci Educ 13:159-66. 10.1187/cbe.CBE-14-03-0054
- Hong L, Page SE. 2004. Groups of diverse problem solvers can outperform groups of high-ability problem solvers. Proc Natl Acad Sci U S A 101:16385-9. 10.1073/pnas.0403723101
- (ed). 2018. Phage Discovery Guide. Howard Hughes Medical Institute, Chevy Chase, Maryland. https://seaphagesphagediscoveryguide.helpdocsonline.com/home.
Article Files
Login to access supporting documents
Data Analysis Recitation Activities Support Better Understanding in SEA-PHAGES CURE(PDF | 248 KB)
Supporting File S1. SEA-PHAGES Recitation Activities - Direct Isolation Student Worksheet.docx(DOCX | 49 KB)
Supporting File S2. SEA-PHAGES Recitation Activities - Direct Isolation Facilitation Guide.docx(DOCX | 45 KB)
Supporting File S3. SEA-PHAGES Recitation Activities - Three-Phase Streak Student Worksheet.docx(DOCX | 149 KB)
Supporting File S4. SEA-PHAGES Recitation Activities - Three Phase Streak Facilitation Guide.docx(DOCX | 149 KB)
Supporting File S5. SEA-PHAGES Recitation Activities - Serial Dilution Student Worksheet.docx(DOCX | 210 KB)
Supporting File S6. SEA-PHAGES Recitation Activities - Serial Dilution Facilitation Guide.docx(DOCX | 268 KB)
Supporting File S7. SEA-PHAGES Recitation Activities - DNA Extraction Student Worksheet.docx(DOCX | 223 KB)
Supporting File S8. SEA-PHAGES Recitation Activities - DNA Extraction Facilitation Guide.docx(DOCX | 456 KB)
Supporting File S9. SEA-PHAGES Recitation Activities - Restriction Enzymes and Gels Student Worksheet.docx(DOCX | 1 MB)
Supporting File S10. SEA-PHAGES Recitation Activities - Restriction Enzymes and Gels Facilitation Guide.docx(DOCX | 1 MB)
Supporting File S11. SEA-PHAGES Recitation Activities-Questions Added to SALG Survey to Assess the RAILS Activities.pdf(PDF | 2 MB)
- License terms

Comments
Comments
There are no comments on this resource.