My Fish Tank: An Active Learning Activity for Bacterial Nitrogen Metabolism
Editor: Mary Mawn
Published online:
Abstract
This active learning activity introduces students to the second part of the nitrogen cycle, nitrification. In terrestrial and aquatic environments, bacteria from the Nitrosomonas and Nitrobacter genera oxidize ammonia first to nitrite (via a hydroxylamine intermediate) and then to nitrate, which is less toxic to animals such as the fish in a fish tank. Nitrification has many practical implications, for example in waste water treatment, but also for those wanting to set up their own fish tank. The activity consists of a pre-class reading, a 50-minute class session, and a home assignment. The class session covers two exercises, each consisting of a group discussion, followed by student reporting, and a compilation of information by the instructor. Students will identify animal nitrogen waste products and fish skin bacteria that are involved in nitrification. Students will also identify the metabolic reactions of the nitrogen cycle, the oxidative state of nitrogen in four metabolic compounds, and the number of electrons transferred through each reaction. The third and final exercise is the take-home assignment, where students write about how they would set up their own fish tank and take care of their fish based on knowledge gained from the in-class exercise.
Primary Image: Nitrification is one of three parts of the nitrogen cycle in both terrestrial and aquatic environments. Nitrification consists of two reactions that first convert ammonia to nitrite and then to nitrate. In the fish tank, this serves to detoxify the water that the fish live in.
Citation
Prüß BM. 2023. My Fish Tank: An Active Learning Activity for Bacterial Nitrogen Metabolism. CourseSource 10. https://doi.org/10.24918/cs.2023.36
Society Learning Goals
Microbiology
- Metabolism
- How are the interactions of microorganisms among themselves and with their environment determined by their metabolic abilities?
- Systems
- How do microorganisms, cellular and viral, interact with both human and non-human hosts in beneficial, neutral, or detrimental ways?
- Impact of Microorganisms
- How can humans utilize and harness microbes and their products?
Lesson Learning Goals
This active learning activity will focus on the process of nitrification, which constitutes one of the three parts of the nitrogen cycle. The cycle encompasses several organisms, including plants and animals; nitrification itself takes place in bacteria. The activity will give students an opportunity to study interactions between bacteria and other organisms, investigate several of the biochemical reactions by the bacteria, and read about nitrification in the applied context of waste water treatment. The conclusion of the exercise is a simple application that students perform by using gained knowledge from the activity to describe how they would set up their own fish tank. The learning objectives align with three of the learning goals from the Microbiology Learning Framework from the American Society for Microbiology (ASM) (see above under Society Learning Goals).
Lesson Learning Objectives
Students will be able to:
- list the nitrogen waste products of different groups of animals.
- identify the two bacteria that live on the skin of fish.
- name the metabolic reactions that the two bacteria perform.
- explain the flow of electrons through the different intermediates of each of the reactions.
- apply knowledge to two applications: waste water treatment and fish tank set up.
Article Context
Course
Article Type
Course Level
Bloom's Cognitive Level
Vision and Change Core Competencies
Vision and Change Core Concepts
Class Type
Class Size
Audience
Lesson Length
Pedagogical Approaches
Principles of How People Learn
Assessment Type
Introduction
Central and intermediary metabolism in bacteria and other organisms follow cycles of nutrients (e.g., carbon, nitrogen, sulfur, etc.), typically consisting of catabolic and anabolic pathways. Catabolic pathways are oxidations that provide the cell with energy. Most anabolic pathways are reductions that provide the cell and often other organisms with building blocks. As one example, the carbon cycle consists of (i) several catabolic pathways that lead to the degradation of sugars and other high energy molecules and provide the cell with energy and (ii) anabolic pathways that use the energy from the sun to fix carbon dioxide and provide sugar building blocks. These reactions do not happen in the same cell or even the same organism. In particular, carbon dioxide fixation is limited to plants and photosynthetic cyanobacteria, most of which fix carbon dioxide via the Calvin cycle, which consists of two light reactions and two photosystems connected by an electron transport chain (photosynthesis; 1). A smaller number of bacteria are capable of using the reverse tricarboxylic acid cycle for carbon dioxide fixation (2). The results of both of these sets of reactions are carbon rich metabolites (e.g., sugars) that are made available to organisms that are not capable of fixing carbon dioxide through the food chain.
The nitrogen cycle is constructed in a similar way. Multiple oxidation and reduction reactions are performed by different organisms to cycle nitrogen. The terrestrial nitrogen cycle can be divided into three primary processes: nitrogen fixation, nitrification, and denitrification (reviewed by 3). Nitrogen fixation is the conversion of atmospheric nitrogen to ammonia, which plants can take up and metabolize. This process is dependent on the metabolic activity of microbes; in particular, bacteria that live in the root nodules of legumes (reviewed by 4). Examples of bacteria that are capable of performing this process include species of the genus Rhizobium or Sinorhizobium. Most of these bacteria are specific for the plant in whose nodules they live (Rhizobium leguminosarum bv. viciae, peas 5; Sinorhizobium meliloti, alfalfa 6). Nitrogen fixation can be introduced to instructors and students using the Three Sisters of Agriculture active learning exercise (7).
Nitrification is the oxidation of ammonia through a hydroxylamine intermediate to nitrite in a first reaction, and then to nitrate in a second reaction. Whether in terrestrial or aquatic environments, this process is performed by bacteria of the genus Nitrosomonas (8) and Nitrobacter (9), respectively. Denitrification is the reverse of this process, the reduction of nitrate to ammonia (compared between ecosystems [10]). Figure 1 compares the nitrogen cycle in terrestrial to aquatic environments. Note that the cycle is shorter in aquatic systems because of the lack of fixation of atmospheric nitrogen at the root nodules of plants.
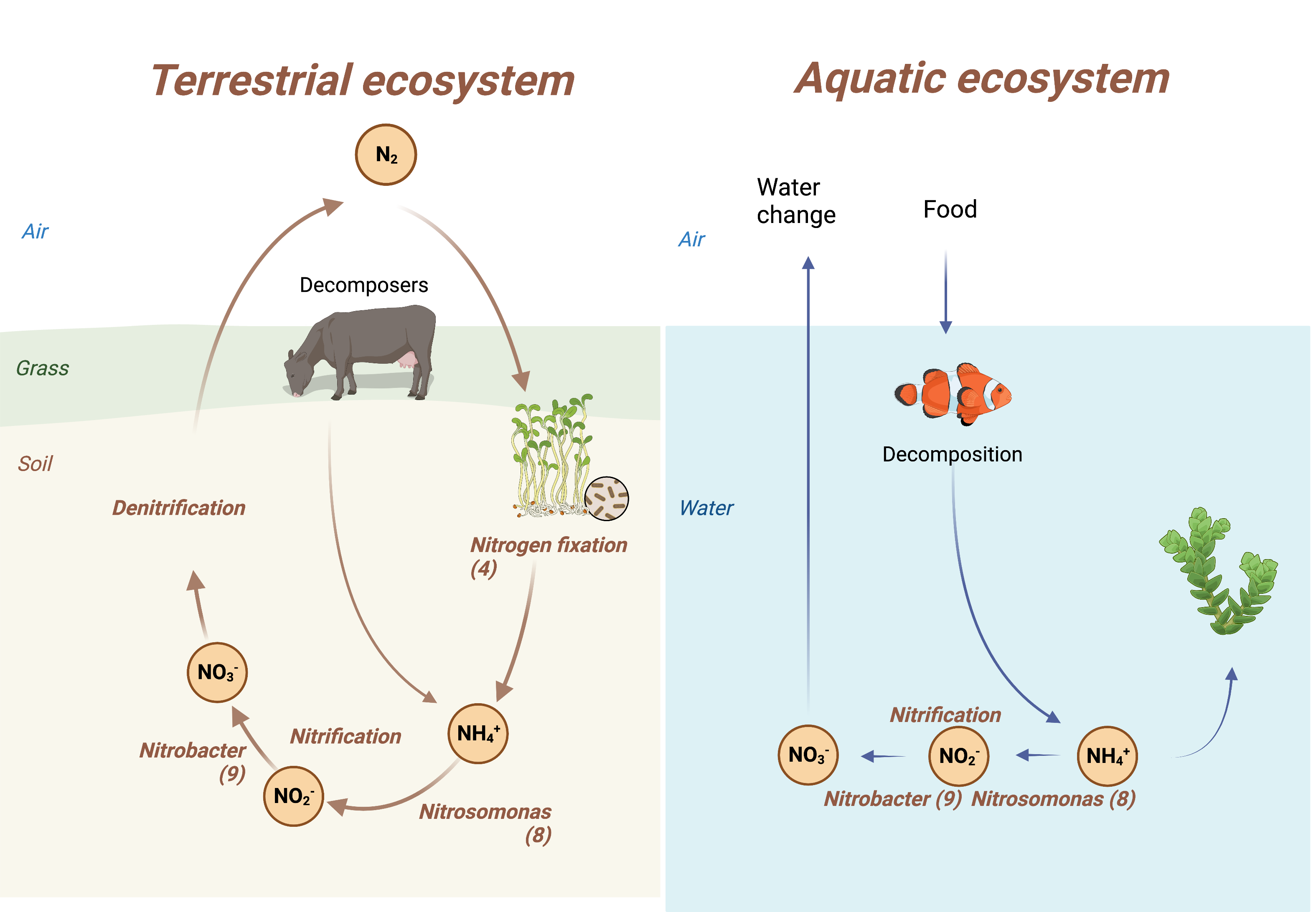
The figure has been constructed on ©BioRender; references for the reactions of nitrogen fixation and nitrification are indicated as part of the figure.
In a tank, fish excrete large amounts of ammonia as an end product of their nitrogen metabolism, which then poisons their ‘breathing’ water. Fish have two types of bacteria on their skin, Nitrosomonas and Nitrobacter. As in terrestrial environments, in this aquatic environment the bacteria metabolize ammonia first to nitrite, which is slightly less toxic than ammonia, and then to nitrate, which is only moderately toxic to the fish. Ammonia can also be removed by plants or algae. All nitrogen compounds (e.g., ammonia, nitrite, and nitrate) can be removed by frequent partial water changes. There are many websites that describe how to keep balance in a fish tank. Note that there are biofilters that can aid the nitrification process in the fish tank. The predominant nitrifying organisms in these filters are ammonia-oxidizing archaea, which accounted for 81 to 86% of nitrogen conversion in in six freshwater aquaria (11). These types of organisms also contribute to nitrification in marine aquatic environments (11, 12).
Structure of the Exercise
In the first part of the exercise, groups of 3 to 4 students are asked to list the nitrogen waste products of different groups of animals, the two bacteria that live on the fish’s skin, and the metabolic reactions that the bacteria perform. Students use an article on LiveAquaria to complete this exercise, discuss their findings for a few minutes within their group, report to the class, and help the instructor to put together a table on the whiteboard that summarizes their outcomes.
The remainder of the in-class session is spent on the second, more advanced part of the activity. The oxidative process of nitrification that the bacteria perform involves the transfer of a large number of electrons. Students are given a research article that helps to explain these reactions (13). The article describes the development of a waste water treatment system that depends on Nitrosomonas europaea and Nitrobacter winogradskyi and is to be used for long term space missions. In addition to interesting information about waste water treatment, the Introduction contains a short paragraph summarizing the stoichiometry of the total reaction sequence from ammonia to nitrate. In class, students are given time for a short group discussion on waste water treatment and electron transfer through the reactions, after which they will report their results to the class. In the process of reporting their results, the instructor gauges the depth of their knowledge, then compiles a short summary of the reactions and the electrons on the whiteboard.
The last component of the lesson is a homework assignment which allows students to apply what they have learned. Students will write a two-page essay on how to set up a fish tank and take care of their own fish. This essay will be based on information from class, but can be supplemented with additional information students can find on the internet.
Intended Audience
This activity targets microbiology majors and minors, as well as students from other areas with an interest in the life sciences. It is possible to teach the first exercise to students at first-year or sophomore level. This part of the exercise helps group members to get to know each other and start with an easy topic. The more advanced second exercise is targeted towards students at junior or senior level, or first-year graduate students. The scientific article will help them develop their knowledge acquisition skills based on primary science tools. This exercise can be used in all three modes of delivery: face to face, online, or HyFlex.
Required Learning Time
Students and instructors will need approximately 30 minutes prior to class to read the Introduction of the research article, find the reaction, and determine the oxidative state of nitrogen in the four nitrogen compounds (13). The class period of 50 minutes will allow for 10 minutes of group discussion and 5 minutes of reporting the results for exercise 1, followed by 20 minutes of group discussion and 10 minutes to complete the reporting for exercise 2. This leaves 5 minutes to explain exercise 3. Students will need an additional 30 minutes to complete the homework exercise 3, and instructors may need 10 minutes to read and grade the exercise (per group).
Prerequisite Student Knowledge
Students should have a general knowledge of central metabolism in bacteria and other organisms. If they do not know what cycling of nutrients is, this description on Treehugger is helpful. There are also multiple video on YouTube, such as this one.
Prerequisite Teacher Knowledge
Content knowledge includes the nitrogen cycle in terrestrial and aquatic systems. This knowledge can be gained from textbooks in microbiology and/or biochemistry, as well as the sources that are indicated above for students. For instructors with an interest that goes beyond the course, the disruption of the nitrogen cycle by anthropogenic activity is described by many research articles, including a paper by Guiry and coworkers (14), which discusses climate change.
Scientific Teaching Themes
Active Learning
This fish tank exercise is clearly structured into three exercises, each of which has a short assessment that can be either formative or summative. Students are given specific scientific literature and are told which parts of the papers they will have to read prior to class. They are also given Figure 1, which summarizes the nitrogen cycle in terrestrial and aquatic ecosystems. In-class time is structured into two individual exercises: both of which start with a student group discussion and a report to the class and end with the instructor’s summarizing the information on a physical or electronic whiteboard. Both the discussions and the report give the activity structure, but they also offer opportunities for student engagement. Students have the opportunity to find scientifically correct information by themselves, only reporting to the entire class after they have agreed on information in the small group.
Students with anxiety may find active learning environments either helpful or harmful, depending on the effectiveness of the active learning exercise (15). This activity was designed such that students can do the active learning exercises alone and turn in reports of the three individual exercises. Hence, students who feel uncomfortable with group work for any reason will have an alternative.
Assessment
The assessment plan contains two pieces of formative assessment. The instructor will listen to students during the breakout sessions and when students report to the class. While constructing the table for the first exercise and the electron flow for the second exercise, the instructor collects information from the students and can assess whether students came to the correct conclusion. The reporting period also gives students the opportunity to assess their own learning in comparison to the class, while still enhancing each other’s learning. The homework assignment, exercise 3, gives students the opportunity to apply their newly gained knowledge to a simple problem that is relevant to nitrification. This is the only piece of summative assessment.
Inclusive Teaching
The opportunity gap between privileged and underprivileged students is often due to a lack of a sense of belonging (16). The multiple ways students can learn throughout this exercise give them the opportunity to learn for themselves first and then be a part of a group. There will be interactions between students within the group and possibly with the instructor during the breakout room discussions. During the reporting period, there will certainly be opportunity for interaction with the instructor. These multiple levels of interaction should make it easier for students to develop a sense of belonging in the class. Another strategy for inclusive teaching is to use empathy (17). An instructor can accommodate individual situations in which students find themselves, demonstrate an interest in their problems, and usually show up early in class to engage students in a variety of topics different from the course.
Lesson Plan
Lesson Overview
The fish tank active learning activity includes a pre-class reading of the introduction of a research article to be done by individual students at home. The in-class session includes two exercises, each consisting of a group discussion followed by a report to the class. The third and final exercise is an applied home assignment. Table 1 guides instructors through these activities.
Table 1. Lesson plan timeline. This table summarizes the different activities. The notes contain tasks to be performed by the students, questions to answer, and tasks to be performed by instructors.
| Activity | Description | Estimated Time | Notes |
|---|---|---|---|
| Preparation for Class | |||
| At home reading |
Instructor and students read the introduction of the research article (8) |
30 min prior to class |
|
|
Class Session (50 min) |
|||
| Student group discussion | Students use Google, YouTube, or other sources to find general information | 10 min |
|
| Student reporting | Students report their findings | 5 min |
|
| Student group discussion | Students discuss the research article (8) | 20 min |
|
| Student reporting | Students report their findings | 10 min |
|
| Instructor | Explains homework | 5 min |
|
| After Class | |||
| Exercise 3 | Students write assignment | 1 h |
|
Pre-Class Preparation
Students should read the Introduction of the research article (13) that is provided to them via their LMS. They will have to find the total reaction in the introduction and determine the oxidative state of the nitrogen in all nitrogen compounds involved. If this is the first time they are learning about nutrient cycling, they may be well advised to read/watch the cycling description on Treehugger and the YouTube video on the nitrogen cycle, as well as Figure 1.
Class Session
The 50-minute class session consists of two rounds of student group discussion and student reporting, covering exercises 1 and 2. Exercise 1 is the retrieval of general information about nitrogen waste products, the two bacteria that live on the skin of fish, and the multiple reactions that these bacteria perform to detoxify the water. Students will form groups of 3 to 4 and can either work in groups within the classroom, or in breakout rooms. They will use an article on LiveAquaria to find the requested information and discuss their findings. At the end of the 10-minute discussion period, one student per group will report their findings to the class. The instructor will use the information to construct Table 2 on the whiteboard. If time permits, the instructor may ask the students how the environment the animals live in impacts the nitrogen waste product. For example, fish are surrounded by water and can afford to expel excess nitrogen into the water as highly water soluble ammonia. Reptiles and birds often live in arid conditions, where water is sparse. They use the poorly water soluble uric acid to expel excess nitrogen. Mammals drink quite a bit of water and use the moderately water soluble urea as a nitrogen excretion product.
Table 2. Assessment table general information. The three questions students are expected to be able to answer during exercise 1 refer to the nitrogen waste products, the two bacteria on the skin of fish, and the metabolic reactions that detoxify water. Answers are indicated. It is advised that instructors construct this table on the whiteboard as students are reporting their findings.
| Question | Sub-Question | Estimated Time | Answer |
|---|---|---|---|
| Which are the nitrogen waste products of these animals? |
|
3 min |
|
| What are the two bacterial species on the skin of fish? |
|
1 min |
|
| What are the two reactions that detoxify water? |
|
1 min |
|
The second exercise is more advanced. Student groups will be given 20 minutes to dissect the total nitrogen reaction from the research article (13), determine the individual reactions, compare their notes on the oxidative state of the nitrogen in each of the nitrogen compounds to those of other students, and calculate the number of electrons that are being transferred during each of the reactions. A student from each group who did not report on exercise 1 will report the findings to the class. The instructor will use the information to write the below reactions, the oxidative state of the nitrogen, and the number of electrons transferred in each of the reactions onto the whiteboard. This takes approximately 10 minutes.
Students should have no problem finding the total reaction in the research article (13):
NH4+ + 1.5 O2 → NO2- + 2H+ + H2ONO2- + 0.5 O2 → NO3-
The reaction includes four compounds that contain nitrogen: ammonia (NH4+), hydroxylamine (H2ONO2-), nitrite (NO2-), and nitrate (NO3-). To calculate the oxidative state of nitrogen in these compounds, students need to know that the net charge of the molecule is always 0. Oxygen counts as -2 and hydrogen counts as +1. Nitrogen can have any oxidative state between -3 and +5. Having this in mind, students should be able to determine that the oxidative state of the nitrogen in ammonia is -3, in hydroxylamine is -1, in nitrate is +3, and in nitrate is +5. That equals a transfer of 8 electrons across the total reaction. Once students dissect the reaction, they will notice that there are actually three reactions; the first two reactions are catalyzed by ammonium monooxygenase and hydroxylamine oxidoreductase in Nitrosomonas europaea, the third is the oxidation of nitrite to nitrate in Nitrobacter winogradskyi. The final electron acceptor is oxygen. Note that the introduction of the article the students studied at home (13) only names these reactions. To identify substrates and products for each of the reactions, students will have to use the internet or textbook. A good reference that explains the details of nitrification could be (18). Below are the three individual reactions:
NH4+ + AH2 + O2 → NH2OH + A + H2O
NH2OH + H2O → NO2- + 5H+ + 4e-
NO2- → NO3-
After the instructor has compiled this information on the whiteboard, there will be 5 minutes left of class time to explain exercise 3, the homework assignment. Students are asked to write a two page essay on how they would set up their own fish tank and detail their plan to maintain their fish.
Post-Class Assignment
The homework assignment that constitutes exercise 3 is to answer two questions: (i) how would you set up your own fish tank? and (ii) what is your plan to maintain your fish? This exercise gives students the opportunity to draw their own conclusions from the knowledge they gained in class. Figure 1 will be provided to them for this exercise. Figure 1 points towards some obvious facts, such as the food the fish will get and the water changes. Students might conclude that too much food will result in too much ammonia, which the bacteria may not be able to clear anymore. Likewise, starting the tank with too many fish will have the same effect. The bacteria have to establish themselves first, not just on the skin but also in the water, to detoxify ammonia. Students should understand that occasional partial water changes are needed. They might conclude that full water changes are less effective, as the bacteria are discarded together with the waste. Figure 1 also shows a plant. A plant in the fish tank uses up ammonia. Students may also come to conclusion that with time there will probably be algae growing on the glass. These too will use up ammonia. This is beneficial to the fish. Figure 1 is constructed in a way that students should be able to draw the above conclusions just from understanding the figure, regardless of whether they ever had a fish tank themselves. If they chose to do so, they can browse the internet for additional information. This might include the biofilters mentioned in the Introduction. The rubric that is provided as Table 3 gives instructors some ideas on how to grade this assignment. This rubric only contains information students can get from the figure. Extra credit can be earned by students who provide additional information.
Table 3. Rubric for exercise 3. The assessment for the home assignment focuses on the four major components of the cycle in the fish tank: food, water, fish, and plant(s). Examples of answers for the different levels of comprehension are given.
| Topic | 0 points | 1 point | 2 points |
|---|---|---|---|
| Food | Students do not comment on food | Students mention that an increase in food will lead to an increase in NH4+ excretion by the fish | Students comment that there may be a concentration of NH4+ that the bacteria may not be able to clear anymore |
| Water | Students do not comment on water | Students comment that water changes are needed | Students conclude that full water changes are less effective because they discard the bacteria |
| Fish | Students do not comment on fish | Students conclude that an increase in the number of fish will lead to an increase in the NH4+ concentration | Students understand that too many fish at the beginning will overwhelm the system before bacteria establish themselves |
| Plant(s) | Students do not comment on plant(s) | Students realize that plants in the tank use up nitrate | Students postulate that with time algae with form and metabolize nitrate just like the plants |
Teaching Discussion
The 400/600 level Bacterial Physiology class is part of the Microbiology program within the College of Agriculture, Food Systems, and Natural Resources. Our Department is home to the North Dakota Agribiome Initiative that is based on the idea of the soil, plant, and animal microbiomes impacting our state’s food production system and contributing to the core of sustainable agriculture (19). One educational aspect of this initiative is that it serves as an example for the influence of bacteria on our society, which connects with the three core concepts.
Many undergraduate student research opportunities arise that keep student excitement and engagement up naturally. Students who engage in research opportunities in this area may already have background knowledge. Still, when teaching a course, especially on a potentially dry topic of physiology, student engagement has to be maintained. Instructors are advised to pay attention to students’ participation during group work or break-out sessions. If needed, an instructor can bring less active students into the discussion. Since students do both individual tasks (prior to class) and group tasks (within-class session), all students are included. Whether the class is taught entirely face to face, entirely online, or HyFlex, groups can be facilitated by moving chairs together or using break out rooms on Zoom. Likewise, the whiteboard can be physical or on Zoom. To enhance student learning, the instructor can follow up on this activity with a lecture on nitrogen metabolism, this may include denitrification.
The assessment plan contains two pieces of formative assessment and one piece of summative assessment. This offers multiple avenues for instructors to assess student learning. The two formative pieces of assessment are exercises 1 and 2 both of which are performed during the in-class session. Both exercises are split into a phase of student group discussion and student reporting (Table 1). Whether the course is taught face to face or online, the instructor is advised to spend 1 or 2 minutes with each of the groups and listen to their discussion. In my class, several students are engaged in the discussion, and the information the students have retrieved has so far always been correct. Likewise, the reported information has always been correct. Students typically respond to my questions quickly, and I make sure it is not always the same student who reports their group’s findings. Sometimes, they struggle with the oxidative state of the nitrogen, and I may have to explain this to them.
For the summative assessment, Table 3 constitutes a rubric which should help instructors to grade the post-class assignment. Instructors should give students the four topics of food, water, fish, and plant(s) and then assess the responses. The learning levels on the Bloom’s taxonomy that can be assessed with the home exercise and the rubric are “synthesis” and “application.”
Adaptations to this exercise can be made for class sizes. For classes of 20 to 30 students, groups of 3 to 4 students work well. For larger classes, instructors would have to either increase the number of students per group or the number of groups. Additional small adaptations can be made regarding the delivery mode. The activity was developed for an active learning classroom. However, only small adaptations are needed for the synchronous online environment, such as the break-out rooms instead of the circles of chairs and the electronic whiteboard as opposed to the one that used to be mounted to the wall of the active learning classroom. In adaptation to lower levels of classes, instructors can omit exercise 2, because this is the most difficult one. Exercises 1 and 3 are easy and can be done by students below a college junior level. As a final note, depending on the learning objectives for the respective courses, this active learning activity can be used in general microbiology or ecology courses. If the general microbiology course is for non-microbiology majors, instructors can limit the exercises to the easier parts 1 and 3.
Acknowledgments
I thank Dr. John McEvoy (NDSU) for helping me with the BioRender figure and Dr. Danielle Condry (NDSU) with the active learning and inclusion sections of the manuscript. The teaching part of my appointment is funded by the College of Agriculture, Food Systems, and Natural Resources. The research part is funded by hatch grant 02438 from USDA/NIFA. I thank the NDSU Gateways ND program for teaching me about backward course design and active learning.
References
- Stirbet A, Lazár D, Guo Y, Govindjee G. 2020. Photosynthesis: Basics, history and modelling. Ann Bot 126:511–537. doi:10.1093/aob/mcz171.
- Rubin-Blum M, Dubilier N, Kleiner M. 2019. Genetic evidence for two carbon fixation pathways (the Calvin-Benson-Bassham cycle and the reverse tricarboxylic acid cycle) in symbiotic and free-living bacteria. mSphere 4. doi:10.1128/msphere.00394-18.
- Stein LY, Klotz MG. 2016. The nitrogen cycle. Curr Biol 26:R94–R98. doi:10.1016/j.cub.2015.12.021.
- Roy S, Liu W, Nandety RS, Crook A, Mysore KS, Pislariu CI, Frugoli J, Dickstein R, Udvardi MK. 2020. Celebrating 20 years of genetic discoveries in legume nodulation and symbiotic nitrogen fixation. Plant Cell 32:15–41. doi:10.1105/tpc.19.00279.
- Yang C, Bueckert R, Schoenau J, Diederichsen A, Zakeri H, Warkentin T. 2017. Symbiosis of selected Rhizobium leguminosarum bv. viciae strains with diverse pea genotypes: Effects on biological nitrogen fixation. Can J Microbiol 63:909–919. doi:10.1139/cjm-2017-0281.
- Santos R, Hérouart D, Sigaud S, Touati D, Puppo A. 2001. Oxidative burst in alfalfa-Sinorhizobium meliloti symbiotic interaction. Mol Plant Microbe Interact 14:86–89. doi:10.1094/MPMI.2001.14.1.86.
- Prüß BM. 2022. The three sisters of agriculture: An active learning activity on symbiotic nitrogen fixation. CourseSource 9. doi:10.24918/cs.2022.40.
- Sedlacek CJ, McGowan B, Suwa Y, Sayavedra-Soto L, Laanbroek HJ, Stein LY, Norton JM, Klotz MG, Bollmann A. 2019. A physiological and genomic comparison of Nitrosomonas cluster 6a and 7 ammonia-oxidizing bacteria. Microb Ecol 78:985–994. doi:10.1007/s00248-019-01378-8.
- Shen Q, Gao J, Liu J, Liu S, Liu Z, Wang Y, Guo B, Zhuang X, Zhuang G. 2016. A new acyl-homoserine lactone molecule generated by Nitrobacter winogradskyi. Sci Rep 6:22903. doi:10.1038/srep22903.
- Seitzinger S, Harrison JA, Böhlke JK, Bouwman AF, Lowrance R, Peterson B, Tobias C, Van Drecht G. 2006. Denitrification across landscapes and waterscapes: A synthesis. Ecol Appl 16:2064–2090. doi:10.1890/1051-0761(2006)016[2064:DALAWA]2.0.CO;2.
- Bagchi S, Vlaeminck SE, Sauder LA, Mosquera M, Neufeld JD, Boon N. 2014. Temporal and spatial stability of ammonia-oxidizing archaea and bacteria in aquarium biofilters. PLoS ONE 9:e113515. doi:10.1371/journal.pone.0113515.
- Ingalls AE, Shah SR, Hansman RL, Aluwihare LI, Santos GM, Druffel ER, Pearson A. 2006. Quantifying archaeal community autotrophy in the mesopelagic ocean using natural radiocarbon. Proc Natl Acad Sci U S A 103:6442–6447. doi:10.1073/pnas.0510157103.
- Cruvellier N, Poughon L, Creuly C, Dussap C-G, Lasseur C. 2016. Growth modelling of Nitrosomonas europaea ATCC® 19718 and Nitrobacter winogradskyi ATCC® 25391: A new online indicator of the partial nitrification. Bioresour Technol 220:369–377. doi:10.1016/j.biortech.2016.08.090.
- Guiry E, Beglane F, Szpak P, Schulting R, McCormick F, Richards MP. 2018. Anthropogenic changes to the Holocene nitrogen cycle in Ireland. Sci Adv 4:eaas9383. doi:10.1126/sciadv.aas9383.
- Cooper KM, Downing VR, Brownell SE. 2018. The influence of active learning practices on student anxiety in large-enrollment college science classrooms. Int J STEM Educ 5:23. doi:10.1186/s40594-018-0123-6.
- Wilson D, Jones D, Bocell F, Crawford J, Kim MJ, Veilleux N, Floyd-Smith T, Bates R, Plett M. 2015. Belonging and academic engagement among undergraduate STEM students: A multi-institutional study. Res High Educ 56:750–776. doi:10.1007/s11162-015-9367-x.
- Dewsbury B, Brame CJ. 2019. Inclusive teaching. CBE Life Sci Educ 18:fe2. doi:10.1187/cbe.19-01-0021.
- Beeckman F, Motte H, Beeckman T. 2018. Nitrification in agricultural soils: Impact, actors and mitigation. Curr Opin Biotechnol 50:166–173. doi:10.1016/j.copbio.2018.01.014.
- Ke J, Wang B, Yoshikuni Y. 2021. Microbiome engineering: Synthetic biology of plant-associated microbiomes in sustainable agriculture. Trends Biotechnol 39:244–261. doi:10.1016/j.tibtech.2020.07.008.
Article Files
Login to access supporting documents

Comments
Comments
There are no comments on this resource.