Introductory Biology courses typically introduce the structure and function of biomolecules such as proteins and nucleic acids. To understand biomolecules fully, students require knowledge of fundamental chemistry concepts such as covalent bonding, intermolecular interactions and hydrophilicity/hydrophobicity (1). Students enter our large (>400 student) course with a notoriously limited conceptual grasp of basic chemistry principles. Our lesson is an activity designed on the principles of POGIL (Process Oriented Guided Inquiry Learning). In 50 minutes, students build their own definitions of the following: polar vs. non-polar covalent bonds, hydrophilicity/hydrophobicity and the nature of hydrogen bonding based simply on the relative electronegativities of oxygen, nitrogen, carbon and hydrogen. We find that this exercise improves students’ understanding of these chemical concepts. Since adopting this activity, students have been better able to understand biomolecular structures and predict interactions between molecules.
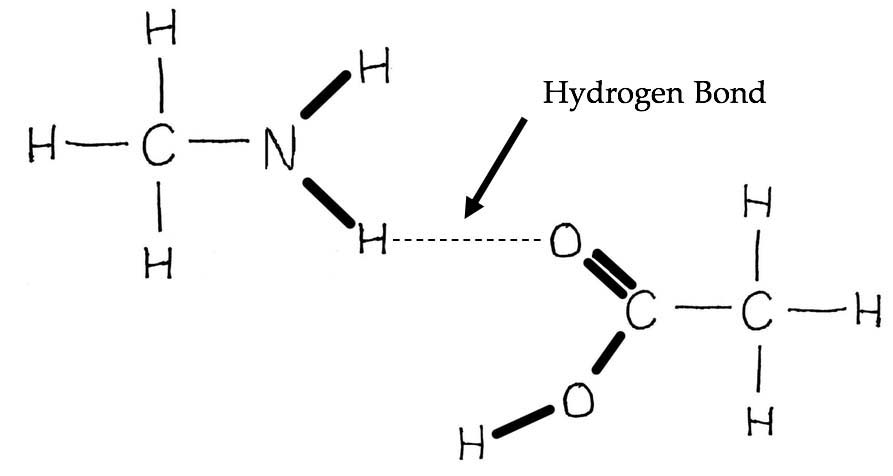
Primary image: Hydrogen Bond. Possible hydrogen bond interaction that can form between two simple organic molecules.

Kevin Law onto Molecular
@
on